2. How has climate changed?

Past climate has varied enormously on a variety of time-scales
Earth’s climate has changed dramatically many times since the planet was formed 4.5 billion years ago. These changes have been triggered by the changing configuration of continents and oceans, changes in the Sun’s intensity, variations in the orbit of Earth, and volcanic eruptions.
Natural variations in the concentrations of greenhouse gases in the atmosphere, the evolution of life and meteorite impacts have also caused climate change in the past. Several million years ago, for example, global average temperature was a few degrees higher than today and warm, tropical waters reached much farther from the equator, resulting in very different patterns of ocean and atmospheric circulation from today.
Over the past million years, Earth’s globally averaged surface temperature has risen and fallen by about 5˚C in ice-age cycles, roughly every 100,000 years or so (Figure 2.1a). In the coldest period of the last ice age, about 20,000 years ago, sea level was at least 120 metres lower than today because more water was locked up on land in polar ice sheets. The last 8,000 years, which includes most recorded human history, have been relatively stable at the warmer end of this temperature range. This stability enabled agriculture, permanent settlements and population growth.
Most past changes in global temperature occurred slowly, over tens of thousands or millions of years. However, there is also evidence that some abrupt changes occurred, at least at regional scales. For example, during the last ice age, temperatures in the North Atlantic region changed by 5°C or more over as little as a few decades, likely due to sudden collapses of Northern Hemisphere ice sheets or changes in ocean currents.
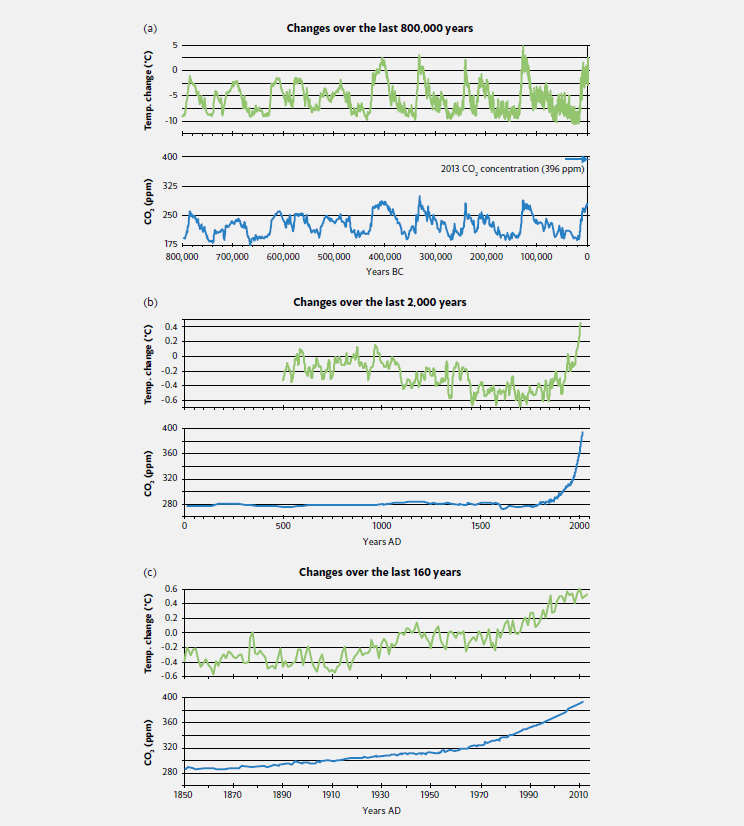
Figure 2.1: Past changes in temperature align with changes in CO2 at a variety of time scales. These graphs show the changes from long‑term average temperature (oC) and average atmospheric CO2 concentration (parts per million) over the last (a) 800,000 years, (b) 2,000 years and (c) 160 years. The temperature changes in (a) are for Antarctica, while for (b) and (c) they are global averages. Source: Compiled from various publicly available data sources as summarised in Box 2.1.
Past records demonstrate that global climate is sensitive to small but persistent influences
Ice-age cycles were initiated by small variations in the rotation of Earth and in its orbit around the sun. These changed the seasonal and latitudinal distribution of solar energy reaching Earth’s surface. Measurements from climate archives such as ice cores (Box 2.1) show that changing temperatures triggered changes to other climate factors such as the concentration of carbon dioxide (CO2) in the atmosphere (Figure 2.1a), amplifying the initial disturbances. During warm periods, the major greenhouse gases CO2 and methane were released into the atmosphere, and receding ice sheets reflected less sunlight to space. These observations confirm that the climate system is sensitive to small disturbances that can be amplified by reinforcing feedback processes. Likewise, the climate system today is sensitive to disturbances from human influences.
Box 2.1: How do we detect climate change?
Identifying temperature change that is global in extent requires frequent observations from many locations around the world. Thermometers, rain gauges and other simple instruments have been used to measure climate variables, starting in the mid-19th century. Over time the quality, variety and quantity of observations has improved. Since the 1970s, sophisticated sensors on earth-orbiting satellites have provided near global coverage of many climate variables. By carefully analysing the data gathered using these techniques (with careful account for changes in instrument types, observational practices, instrument locations and urban areas) it has been possible to map the distribution of temperature and other climate changes since the late 19th century.
To study climate changes that occurred before direct measurements were made, scientists use indirect evidence from other sources that record a climate signal. These include climate signals encoded in the composition of ice cores, corals, sediments in oceans and lakes, and tree rings. All these records are laid down sequentially over time as an organism grows or as sediments accumulate. Ice cores from polar ice sheets, which are built from snow laid down over tens to hundreds of thousands of years, provide records of both past CO2 and temperature. As the snow transforms into ice, it traps air in sealed bubbles that provide a sample of past atmospheric composition, while the ratio of stable isotopes of either oxygen or hydrogen in the water molecule is related to the temperature at the time when the snow fell. More recent historical changes can be identified by analysing written and pictorial records, for example of changes in glacier extent.
Box 2.2: Has climate warming recently stopped?
According to most estimates, the rate of average surface warming has slowed since 2001, despite ongoing rises in greenhouse gases. This slowdown is consistent with known climate variability. Indeed, decades of little or no temperature trend can be seen throughout the last century, superimposed on the long-term warming trend.
Two main factors have contributed to the most recent period of slowed surface warming. First, decadal variability in the ocean-atmosphere system has redistributed heat in the ocean, especially in the eastern and central Pacific. This has caused warming at depth and cooling of surface waters and the lower atmosphere in this region. Second, several temporary global cooling influences have come into play including unusually weak solar activity (Box 3.1), increased aerosol production, and volcanic activity.
None of these influences is likely to continue over the long term. Moreover, despite the slowdown in warming at the surface, there have been continuing increases in heat extremes and in the heat content of the oceans, as well as rising sea levels, shrinking Arctic sea-ice, and ongoing melt of ice sheets and glaciers. Some models predict that, when the current slowdown ends, renewed warming will be rapid.
Global average temperatures have increased over the past century
Climate and sea level were relatively stable over thousands of years of recorded human history up to the 19th century, although with some variations (Figure 2.1b). However, globally averaged near-surface air temperature rose by around 0.8°C between 1850 and 2012 (Figure 2.1c). The rate of warming increased in the mid-1970s, and each of the most recent three decades has been warmer than all preceding decades since 1850. The last decade has been the warmest of these. Satellite observations and direct measurements also show warming in the lower atmosphere over the past three decades. In contrast, the atmosphere above about 15 km elevation (the stratosphere) has cooled over this time.
The temperature of the oceans has also risen. More than 90% of the total heat accumulated in the climate system between 1971 and 2010 has been stored in the oceans. The greatest ocean warming has taken place close to the surface, with the upper 75 m of the ocean warming by an average of 0.11°C each decade between 1971 and 2010.
Changes are evident in many parts of the climate system
Changes consistent with an increase in global temperature have been observed in many other components of the climate system.
- Mountain glaciers have been shrinking and contributing to global sea-level rise since about 1850. Melting accelerated significantly in the 1990s.
- The Greenland and West Antarctic Ice Sheets have both lost ice since 1990, further contributing to sea-level rise as discussed in Question 6. This is from increased discharge of ice into the ocean, and also increased surface melting in Greenland. The rate of loss from Greenland appears to be increasing.
- The area of the Arctic Ocean covered by sea ice has decreased significantly since 1987 throughout the year and particularly in summer. The thickness of the ice has also decreased by more than 30% over the last 30 years.
- In the Southern Ocean, there are strong regional differences in the changes to areas covered by sea ice, but a small increase in total coverage, driven by shifts in winds and ocean currents in a warming Southern Ocean. Strengthening circumpolar winds around Antarctica have also been linked in part to thinning of the ozone layer.
- The amount of water vapour in the atmosphere has increased since the 1980s, which is consistent with warmer air (Box 1.3).
- The surface of the ocean in rainy parts of the world is becoming less salty, which is consistent with freshwater dilution from increased rainfall.
- Some ocean currents have changed in response to changes in surface winds, ocean temperature and ocean saltiness. The changes include a southward shift of the Antarctic Circumpolar Current and increasing southward penetration of the East Australian Current.
- An increasing number of plants and animals, on land and in the oceans, are undergoing shifts in their distribution and lifecycles that are consistent with observed temperature changes.
There are regional differences to climate change including within Australia
Over the past 100 years, temperature has increased over almost the entire globe; the rate of increase has been largest in continental interiors (Figure 2.2). The average surface temperatures over the Australian continent and its surrounding oceans have increased by nearly 1°C since the beginning of the 20th century (Figure 2.3). Seven of the ten warmest years on record in Australia have occurred since 2002. However there are differences across Australia with some regions having warmed faster and others showing relatively little warming (Figure 2.3 right).
Since the mid 1990s there have been significant increases in wet season rainfall over northwest Australia (Figure 2.4 left), a declining trend in southwest Australia, and a 15% decline in late autumn and early winter rainfall in the southeast (Figure 2.4 right).
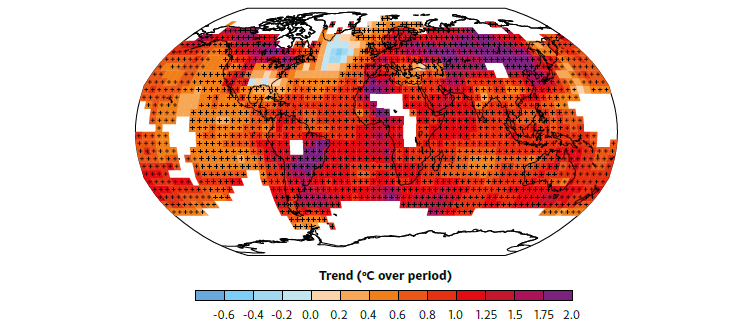
Figure 2.2: Surface temperature has increased across most of the world since 1901. This map shows the distribution of the average temperature change between 1901 and 2012. Adapted from IPCC (2013), Fifth Assessment Report, Working Group 1, Figure 2.21.
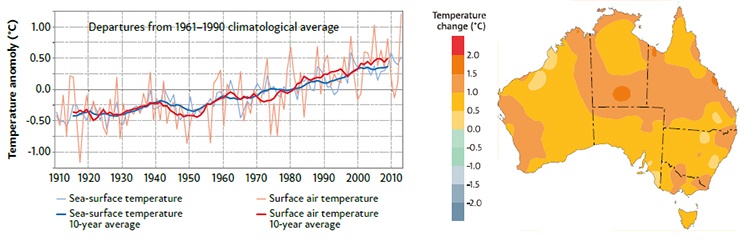
Figure 2.3: Temperature has risen over Australia and in the surrounding ocean since the beginning of the 20th century, although there are regional differences. Plot on left shows deviations from the 1961–1990 average of sea surface temperature and temperatures over land in the Australian region; map on right shows distribution of annual average temperature change across Australia since 1910. Adapted from BOM/CSIRO State of the Climate 2014.
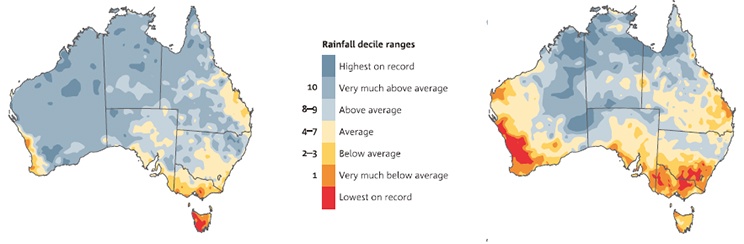
Figure 2.4: Recent rainfall in northern Australia has been higher than average during the northern wet season, and in southern Australia it has been drier during the southern wet season. The maps show the relative ranking (in 10% increments) of rainfall from July 1995 to June 2014 compared with the average since 1900 for (left) northern Australian wet season (Oct–Apr) and (right) southern Australian wet season (Apr–Nov). Adapted from BOM/CSIRO State of the Climate 2014.
1. What is climate change?

Climate change is a change in the pattern of weather, and related changes in oceans, land surfaces and ice sheets, occurring over time scales of decades or longer
Weather is the state of the atmosphere—its temperature, humidity, wind, rainfall and so on—over hours to weeks. It is influenced by the oceans, land surfaces and ice sheets, which together with the atmosphere form what is called the ‘climate system’. Climate, in its broadest sense, is the statistical description of the state of the climate system.
Climate change is a change in the statistical properties of the climate system that persists for several decades or longer—usually at least 30 years. These statistical properties include averages, variability and extremes. Climate change may be due to natural processes, such as changes in the Sun’s radiation, volcanoes or internal variability in the climate system, or due to human influences such as changes in the composition of the atmosphere or land use.
Weather can be forecast with considerable skill up to about a week in advance. Short term fluctuations in climate, such as droughts, can be predicted with limited skill from season to season. In contrast, changes in the long-term statistics of the climate system (climate change) can be predicted if caused by long-term influences that are known or predictable (Box 1.1).
Box 1.1: If weather can only be forecast about a week in advance, how can we determine future climate?
The challenges of predicting weather and climate are very different. Predicting the weather is like predicting how a particular eddy will move and evolve in a turbulent river: it is possible over short time scales by extrapolating the previous path of the eddy, but eventually the eddy is influenced by neighbouring eddies and currents to the extent that predicting its exact path and behaviour becomes impossible. Similarly, the limit for predicting individual weather systems in the atmosphere is around 10 days. On the other hand, predicting climate is like predicting the flow of the whole river. It requires a consideration of the major forces controlling the river such as changes in rainfall, the operation of dams, and extraction of water. Projections of human-induced climate change over decades to centuries are possible because human activities have predictable effects on the future atmospheric composition, and in turn a predictable effect on climate.
Climate is determined by many factors that influence flows of energy through the climate system, including greenhouse gases
Energy from the Sun is the ultimate driver of climate on Earth. The solar energy received by Earth depends on how much the Sun emits and the distance between Earth and the Sun. Part of this sunlight is reflected directly back to space by the atmosphere, clouds, and land, ice and water surfaces. Aerosols (tiny particles in the atmosphere, some coming from human activities) can increase the reflection of sunlight.
Eventually the solar energy absorbed by Earth is returned to space as infrared (heat) radiation. In the process it interacts with the whole climate system—atmosphere, oceans, land surfaces and ice sheets. The flows of radiation in the atmosphere (Figure 1.1) are very important in determining climate. The main gases that make up the atmosphere, nitrogen and oxygen, do not interact with infrared radiation. However, certain gases present in smaller quantities absorb infrared radiation flowing upwards from Earth’s surface and re-radiate it in all directions, including back downwards. By doing this they impede the outward flow of infrared energy from Earth to space. This is called the ‘greenhouse effect’, and the gases that cause it by interacting with infrared radiation are called greenhouse gases. The most important are water vapour, carbon dioxide (CO2) and methane. The greenhouse effect was identified more than a century ago; Earth’s surface would be about 33°C cooler without it, so it keeps Earth habitable.
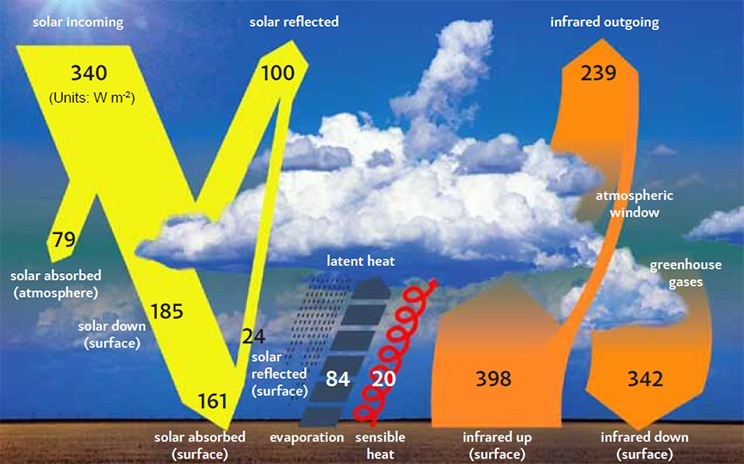
Figure 1.1: The rates at which energy enters the Earth system from the Sun, and leaves the system, approximately balance on average globally. Energy absorbed at the surface is transferred to the atmosphere via infrared radiation, conduction of sensible heat, and evaporation of water whose latent heat is released later when the water condenses again. Energy leaves the system mostly via infrared radiation from the atmosphere. The arrows show global average energy transfer rates in units of Watts per square metre. With more greenhouse gases in the atmosphere, but no other changes, the system must reach a higher temperature to maintain balance. Adapted from IPCC (2013), Fifth Assessment Report, Working Group 1, Figure 2.11.
Changes in climate can occur through both natural and human-induced causes
Global climate varies naturally over time scales from decades to thousands of years and longer. These natural variations can originate in two ways: from internal fluctuations that exchange energy, water and carbon between the atmosphere, oceans, land and ice, and from external influences on the climate system, including variations in the energy received from the sun and the effects of volcanic eruptions.
Human activities can also influence climate by changing concentrations of CO2 and other greenhouse gases in the atmosphere (Box 1.2), altering the concentrations of aerosols and altering the reflectivity of Earth’s surface by changing land cover.
Box 1.2: How do human activities enhance the ‘greenhouse effect’?
Today, human activities are directly increasing atmospheric concentrations of CO2, methane and nitrous oxide, plus some chemically manufactured greenhouse gases such as halocarbons (Question 3). These human generated gases enhance the natural greenhouse effect and further warm the surface. In addition to the direct effect, the warming that results from increased concentrations of long-lived greenhouse gases can be amplified by other processes. A key example is water vapour amplification (Box 1.3). Human activities are also increasing aerosols in the atmosphere, which reflect some incoming sunlight. This human-induced change offsets some of the warming from greenhouse gases.
A disturbance to the climate system can trigger further changes that amplify or damp the initial disturbance
There are close connections between temperature, atmospheric water vapour, the extent of polar ice sheets and the concentrations of long-lived greenhouse gases (especially CO2) in the atmosphere.
When one of these is disturbed, the others react through ‘feedback’ processes that may amplify or dampen the original disturbance. These feedbacks occur on a wide range of time scales: those involving the atmosphere are typically rapid, while those involving deep oceans and ice sheets are slow and can cause delayed responses.
An example of a rapid feedback is the role of water vapour as explained in Box 1.3. An example of a slow feedback is the ice age cycles that have taken place over the past million years, triggered by fluctuations in Earth’s rotation and orbit around the sun. These fluctuations changed the distribution of solar radiation received by Earth, which caused temperatures to change, in turn inducing changes in ice sheets and carbon cycling that together amplified the temperature response. (Question 2).
Box 1.3: If water vapour is the most important greenhouse gas, why all the fuss about CO2?
Water vapour accounts for about half the natural greenhouse effect. Its concentrations in the atmosphere are controlled mainly by atmospheric temperatures and winds, in contrast with the concentrations of other greenhouse gases which are directly influenced by human-induced inputs of these gases to the atmosphere. When global average atmospheric temperatures rise, global water vapour concentrations increase, amplifying the initial warming through an enhanced greenhouse effect. In this way, human activity leads indirectly to increases in water vapour concentrations.
The reality of the water vapour feedback is supported by recent observations and analyses. Increased water vapour concentrations have been observed and attributed to warming, and this feedback approximately doubles the sensitivity of climate to human activities.
The science of climate change: About

Working Group:
- Lisa Alexander
- Ian Allison AO (co-chair)
- Michael Bird
- John Church FAA FTSE
- Matthew England FAA
- Jean Palutikof
- Michael Raupach FAA FTSE (co-chair)
- Steven Sherwood
- Susan Wijffels
Oversight Committee:
- Graham Farquhar AO FAA FRS
- Roger Gifford
- Andrew Gleadow FAA
- Kurt Lambeck AO FAA FRS
- Trevor McDougall FAA FRS
- Graeme Pearman AM FAA FTSE
- Steve Rintoul FAA
- John Zillman AO FAA FTSE (chair)
Acknowledgments:
- Hedda Ransan-Cooper (scientific editing)
- Stephen Pincock (communication consultant)
- Louise Bell
- Jules Kajtar
- Jana Sillmann (figure preparation)
©2015 Australian Academy of Science, GPO Box 783, Canberra ACT 2601, all rights reserved. Selected passages, tables or diagrams may be reproduced, provided the source is acknowledged.
The science of climate change: summary

Earth’s climate has changed over the past century. The atmosphere and oceans have warmed, sea levels have risen, and glaciers and ice sheets have decreased in size. The best available evidence indicates that greenhouse gas emissions from human activities are the main cause. Continuing increases in greenhouse gases will produce further warming and other changes in Earth’s physical environment and ecosystems.
The science behind these statements is supported by extensive studies based on four main lines of evidence:
- Physical principles established more than a century ago tell us that certain trace gases in the atmosphere, such as carbon dioxide (CO2) and water vapour, restrict the radiant flow of heat from Earth to space. This mechanism, known as the ‘greenhouse effect’, keeps Earth’s surface and lower atmosphere considerably warmer than they would otherwise be. The gases involved are called ‘greenhouse gases’. An increase in greenhouse gas concentrations raises the temperature of the surface.
- The record of the distant past(millions of years) tells us that climate has varied greatly through Earth’s history. It has, for example, gone through ten major ice age cycles over approximately the past million years. Over the last few thousand years of this period, during which civilisations developed, climate was unusually stable. Evidence from the past confirms that climate can be sensitive to small persistent changes, such as variations in Earth’s orbit.
- Measurements from the recent past (the last 150 years) tell us that Earth’s surface has warmed as atmospheric concentrations of greenhouse gases increased through human activities, and that this warming has led to other environmental changes. Although climate varies from decade to decade, the overall upward trend of average global surface temperature over the last century is clear.
- Climate models allow us to understand the causes of past climate changes, and to project climate change into the future. Together with physical principles and knowledge of past variations, models provide compelling evidence that recent changes are due to increased greenhouse gas concentrations in the atmosphere. They tell us that, unless greenhouse gas emissions are reduced greatly and greenhouse gas concentrations are stabilised, greenhouse warming will continue to increase.
This document aims to summarise and clarify the current scientific understanding of climate change by answering nine key questions.
1 What is climate change?
The term ‘climate’, in its broadest sense, refers to a statistical description of weather and of the related conditions of oceans, land surfaces and ice sheets. This includes consideration of averages, variability and extremes. Climate change is an alteration in the pattern of climate over a long period of time, and may be due to a combination of natural and humaninduced causes.
2 How has climate changed?
Global climate has varied greatly throughout Earth’s history. In the final decades of the 20th century, the world experienced a rate of warming that is unprecedented for thousands of years, as far as we can tell from the available evidence. Global average temperature rise has been accompanied by ongoing rises in ocean temperatures, ocean heat storage, sea levels and atmospheric water vapour. There has also been shrinkage in the size of ice sheets and most glaciers. The recent slowdown in the rate of surface warming is mainly due to climate variability that has redistributed heat in the ocean, causing warming at depth and cooling of surface waters. Australia’s climate has warmed along with the global average warming.
3 Are human activities causing climate change?
Human activities are increasing greenhouse gas concentrations in the atmosphere. This increase is extremely likely to have caused most of the recent observed global warming, with CO2
4 How do we expect climate to evolve in the future?
If greenhouse gas emissions continue to grow rapidly, it is expected that, by 2100, the global average air temperature over the Earth’s surface will warm by around 4°C above mid-19th century temperatures. There are many likely ramifications of this warming. However, if emissions are reduced sufficiently rapidly, there is a chance that global average warming will not exceed 2°C and other impacts will be limited.
5 How are extreme events changing?
Since the mid-20th century, climate change has resulted in increases in the frequency and intensity of very hot days and decreases in very cold days. These trends will continue with further global warming. Heavy rainfall events have intensified over most land areas and will likely continue to do so, but changes are expected to vary by region.
6 How are sea levels changing?
Sea levels have risen during the 20th century. The two major contributing factors are the expansion of sea water as it warms, and the loss of ice from glaciers. Sea levels are very likely to rise more quickly during the 21st century than the 20th century, and will continue to rise for many centuries.
7 What are the impacts of climate change?
Climate change has impacts on ecosystems, coastal systems, fire regimes, food and water security, health, infrastructure and human security. Impacts on ecosystems and societies are already occurring around the world, including in Australia. The impacts will vary from one region to another and, in the short term, can be both positive and negative. In the future, the impacts of climate change will intensify and interact with other stresses. If greenhouse gas emissions continue to be high, it is likely that the human-induced component of climate change will exceed the capacity of some countries to adapt.
8 What are the uncertainties and their implications?
There is near-unanimous agreement among climate scientists that human-caused global warming is real. However, future climate change and its effects are hard to predict accurately or in detail, especially at regional and local levels. Many factors prevent more accurate predictions, and some uncertainty is likely to remain for considerable time. Uncertainty in climate science is no greater than in other areas where policy decisions are routinely taken to minimise risk. Also, the uncertainty means that the magnitude of future climate change could be either greater or less than present-day best estimates.
9 What does science say about options to address climate change?
Societies, including Australia, face choices about how to respond to the consequences of future climate change. Available strategies include reducing emissions, capturing CO2, adaptation and ‘geoengineering’. These strategies, which can be combined to some extent, carry different levels of environmental risk and different societal consequences. The role of climate science is to inform decisions by providing the best possible knowledge of climate outcomes and the consequences of alternative courses of action.
The science of climate change: Foreword

The purpose of this booklet is to provide an understanding, based on our present scientific knowledge, of some key questions about climate change.
It is an extensively revised update of a similarly titled Academy publication in 2010 that summarised the state of knowledge at that time. It has been prepared by a broadly-based Working Group of Australian climate scientists with review and guidance provided by an Oversight Committee composed of Academy Fellows and the former Chair of the Academy’s National Committee for Earth System Science.
Along with its sister Academies, the Australian Academy of Science has played an active role in assessing the science of climate change since the 1970s. The Academy recognises the role of the Intergovernmental Panel on Climate Change (IPCC) as the mechanism for the international scientific assessment of climate change science, impacts and response strategies. However, it believes that it is important that Australian climate scientists explain the science, including its uncertainties and implications, to the Australian community in simpler terms than can be found in most of the IPCC reports.
The Working Group who prepared this update was led by Professor Michael Raupach FAA FTSE and Dr Ian Allison AO with special support, in the later stages, from Professor Steven Sherwood. The views presented in the answers to the nine key questions were carefully reviewed by an Oversight Committee and 12 independent climate scientists* who agreed to help with the preparation of this document. The role of the Oversight Committee was to make sure that all reasonable review comments were properly considered by the Working Group in preparing their final text. While the reviewers provided more than 600 individual comments on the penultimate draft, neither they nor the Oversight Committee are responsible for the final wording of the detailed answers that represent the views of the expert members of the Working Group.
Nevertheless the summary on pages 4 and 5 represents the fully agreed views of both the Oversight Committee and the Working Group. It has been endorsed by the Academy as a balanced, objective and authoritative summary of the current state of knowledge of the science of climate change.
As in all areas of active science, uncertainties remain. However, enormous scientific progress has been made in our understanding of climate change and its causes and implications. Since 2010, the IPCC has prepared a new international assessment with the active involvement of many Australian researchers, including several members of the Academy Working Group. This Q&A update is thus well informed by recent international developments in the science as well as the most recent work by our own scientists on peculiarly Australian aspects of the climate change problem.
As the summary states, ‘Societies, including Australia, face choices about how to respond to the consequences of future climate change.’ It is incumbent on society to consider these choices.
I wish to thank all the members of the Working Group and Oversight Committee (whose names are listed on the back cover) for their painstaking work in the preparation of this update. I also acknowledge the assistance of the reviewers and others who helped with this update. The Academy is especially grateful to the Department of the Environment, which provided the financial support for the preparation and publication of this document.
On behalf of the Academy, I am pleased to commend the information in the following pages to all those who are looking for authoritative answers to the key questions we are all asking about the science of climate change.
Andrew Holmes AM PresAA FRS FTSE
President
Australian Academy of Science
*In addition to multi-stage review carried out by the Oversight Committee, the penultimate draft of this document was reviewed by Dr G Ayers FTSE, Dr I G Enting, Professor D Griggs FTSE, Professor D Karoly, Mr WR Kininmonth, Professor M J Manton FTSE, Dr K G McCracken AO FAA FTSE, Professor N Nicholls, Dr N Smith FTSE and three anonymous reviewers.
The education revolution that drive science and builds nations

Nobel Laureate Brian Schmidt speaking at the 2011 Prime Minister’s Prizes for Science Awards Dinner at Parliament House on 12 October 2011
He came to this country from the US partly because of its unique position and facilities. But Australia has matured since then, says our newest Nobel Laureate, astronomer Brian Schmidt. He is optimistic about the future for Australian science and the contribution it can make to improving lives, in this country and the world. Here is an edited version of the speech he gave at the presentation of the 2011 Prime Minister’s Awards for Science.

Despite my accent, I have lived in Canberra longer than anywhere else in my life. A lot has changed in the past 17 years. When I arrived in Australia in 1994, it was a well-off country separated by vast oceans from the rest of the world. Today, Australia is one of the world’s wealthiest countries, gateway to the fastest growing part of the world economically, Asia.
It has come of age. The world is rapidly changing, and Australia is in a unique position to shape its future for the century ahead.
Eleven years ago I stood up on this podium—as the recipient of the first Malcolm McIntosh Prize for Physical Scientist of the Year—in complete wonderment of the moment. I feel that same wonderment tonight, as we salute Australia’s finest scientists and science educators
When I came to this country, Australia didn’t have a science awards night dedicated to its own researchers. Now, we celebrate our nation’s best scientists and educators on our own terms.
The Malcolm McIntosh Prize was, in fact, the first award I received for my work on the accelerating Universe, setting off a progression which culminated in last week’s Nobel Prize announcement. It is a sign of our nation’s confidence—and important to me—that my home country was able to recognise my part in this discovery first.
I often hear it said that Science and Education policy never won an election. But nations rise and nations fall on the outcomes of science and education. Improvements in our lives are largely due to technology powered by science and education. I guess the lack of political acknowledgement of this is because science and education do not run on a three-year cycle. It takes decades for policies to run their course, but they provide a similarly long legacy.

The policy makers of this generation have a unique opportunity to shape the long term prosperity of this country. Using the opportunities that arise from a prosperous, agile economy, Australia can ensure its future prosperity in a rapidly changing world through a strategic vision of and investment in education, science and technology.
The education part of this triumvirate is straightforward. Australia needs a workforce educated commensurate with its wealth—to put it simply, the world’s best educated workforce. This is the engine of future prosperity.
It should not be surprising that my high school education was sensational. I grew up in Alaska during the oil boom. Alaska paid teachers based on their ability, and paid them exceedingly well, relatively speaking. Among my teachers was a man with a PhD in chemistry from the Massachusetts Institute of Technology. He was also my cross-country running coach.
We should be smart and learn from tonight’s award recipients, but ultimately, as in Alaska, it will cost money, and it will take time. This, however, has to be at the top of our agenda. And, in relative terms, it really isn’t that expensive—12 years of good education provides a 50-year legacy. So, Prime Minister, I applaud the beginning of the education revolution—but let the revolution continue.
Science is the building block of future technological breakthroughs. Basic science research creates revolutionary new ideas. It’s a messy process, but it is the process that has taken our world from the Dark Ages to our current prosperity. Remember that the 2009 Prime Minister’s Prize for Science recipient John O’Sullivan started out by trying to discover evaporating black holes. He never found any of them, but ended up helping to invent the Wi-Fi system we all now use. And the royalties flowing back to Australia from his work are just the beginning. His achievement has increased productivity not just Australia, but around the world.
Tonight we honour Professors Ezio Rizzardo and David Solomon who have used their basic research in polymers to open a whole new way of making innovative products. Again significant royalties will flow from their inventions back to CSIRO. Professor Solomon was also largely responsible for Australia’s plastic banknotes. So science really does make money—directly!
Scientific research thrives in world-class institutions. Australia should strive to strengthen its universities, and also to ensure that CSIRO remains the unique research institution it is. I can see nothing but good emerging from working towards having at least one university in the top 10 internationally, and three in the top 50.
Then there is the process of taking the science and technology to market. This traditionally has been hard. Australian companies have found it difficult to capitalise in our small domestic market—in both senses of the word. But this is an area in which the world order is changing. If Australia works with partners on a more international basis, it will be better able to transform its good ideas into goods and services in the global marketplace.

Working internationally is challenging for governments—a posture here, a step there. Progress is painfully slow. But for scientists, it comes naturally. We routinely work together in the pursuit of knowledge. So science can be a conduit to take Australian industry to the world.
For example, over the past 5 years, with the support of CSIRO and the Commonwealth Government, Australia has pulled out all stops in putting in a superb bid to host the Square Kilometre Array Telescope in Western Australia. This next-generation radio telescope will enable astronomers around the world to make fundamental discoveries of our Universe. But it will also facilitate opportunities for Australian companies to work with their European, Asian and North American counterparts—creating linkages everywhere.
But the Square Kilometre Array is only one of many such international projects. Not all, of course, will be based in Australia, and not all have equal merit. But involvement in a portfolio of such projects can provide a wealth of scientific and industrial opportunities within the international landscape.
The future for Australia is indeed bright—but it is not guaranteed. Capitalising on Australia’s opportunities will not just happen, it requires strategic science and education policies that adapt to a changing world. And Australians will have to be willing to make significant changes in how they go about their business.
But we have all the ingredients for success:
- great teachers, such as science teaching prize winners Brooke Topelberg and Jane Wright, whom we celebrate tonight and from whom future education policy can learn; and
- great scientists, such as science prize winners Min Chen, Stuart Wyithe, Ezio Rizzardo and David Solomon, who enrich our world with new ideas.
While I cannot predict if tonight’s award recipients are future Nobel Prize winners in-the-making, it matters not. Their work requires no further validation. We, as Australians, tonight celebrate the profound contributions of our science prize laureates to our society.
I congratulate this year’s science prize winners, and hope their award is as inspiring and enabling as the Malcolm McIntosh prize has been to me over the past 11 years.
‘Still no Mawson’: Frank Stillwell’s Antarctic diaries 1911–13



In January 1913, Frank Stillwell and 14 other men waited anxiously at Main Base in Antarctica for three overdue men.
All of the summer’s field parties had returned safely to the Cape Denison hut—except for Douglas Mawson’s.
‘Another day and no Mawson’, wrote Stillwell on 21 January. ‘Still no Mawson’, he wrote five days later. ‘The most optimistic among us now are beginning to have fears …’. By February it seemed clear that the missing men had all died.
Frank Stillwell’s diaries reveal everyday life in the men’s isolated hut in Antarctica, with near-poisonings and the tragedy of two men’s deaths.
The diaries are now held by the Academy. With the support of Geoscience Australia and the Geological Society of Australia, the diaries have been published in full in this book. The year 2012 was a centenary year for the 1911–14 Australasian Antarctic Expedition, and this book is a fitting celebration of Australia’s first scientific expedition to Antarctica.
The book is available in Canberra from the National Library of Australia bookshop, ANU Co-op Bookshop, Smith’s Alternative Bookshop and Paperchain Bookstore.
(edited by Bernadette Hince)
Nobel Australians

2025 Nobel Prize in Chemistry
Professor Richard Robson FAA FRS shared with Professor Susumu Kitagawa FRS and Professor Omar M. Yaghi ‘for the development of metal–organic frameworks’.
2017 Nobel Peace Prize
International Campaign to Abolish Nuclear Weapons (ICAN) ‘for its work to draw attention to the catastrophic humanitarian consequences of any use of nuclear weapons and for its ground-breaking efforts to achieve a treaty-based prohibition of such weapons’. ICAN was launched in Melbourne in 2007.
2011 Nobel Prize in Physics
Professor Brian Schmidt AC FAA FRS along with two American researchers, Professor Saul Perlmutter and Adam Riess, ‘for the discovery of the accelerating expansion of the Universe through observations of distant supernovae’.
2009 Nobel Prize in Physiology or Medicine
Professor Elizabeth Blackburn AC FAA FRS, with Professor Carol Greider and Professor Jack Szostak ‘for the discovery of how chromosomes are protected by telomeres and the enzyme telomerase’.
2005 Nobel Prize in Physiology or Medicine
Professor Barry James Marshall AC FAA FRS and Dr Robin Warren AC FAA ‘for their discovery of the bacterium Helicobacter pylori and its role in gastritis and peptic ulcer disease’.
Download a poster
2003 Nobel Prize in Literature
Professor John Maxwell Coetzee FAHA ‘who in innumerable guises portrays the surprising involvement of the outsider’.
1996 Nobel Prize in Physiology or Medicine
Professor Peter Charles Doherty AC FAA FRS shared with Rolf Martin Zinkernagel AC FAA ‘for their discoveries concerning the specificity of the cell mediated immune defence’.
Front of poster
Back of poster
1975 Nobel Prize in Chemistry
Professor Sir John Warcup Cornforth AC FAA FRS shared with Professor Vladimir Prelog FRS ‘for their work on the stereochemistry of enzyme-catalyzed reactions’.
1973 Nobel Prize in Literature
Patrick Victor Martindale White ‘for an epic and psychological narrative art which has introduced a new continent into literature’.
1970 Nobel Prize in Physiology or Medicine
Sir Bernard Katz FRS shared with Professor Ulf von Euler and Julius Axelrod ‘for their discoveries concerning the humoral transmittors in the nerve terminals and the mechanism for their storage, release and inactivation’.
1964 Nobel Prize in Physics
Professor Aleksandr Mikhailovich Prokhorov shared with Charles Hard Townes and Nicolay Gennadiyevich Basov ‘for fundamental work in the field of quantum electronics, which has led to the construction of oscillators and amplifiers based on the maser-laser principle’. Prokhorov was born in Australia but was educated and worked in the Soviet Union.
1963 Nobel Prize in Physiology or Medicine
Sir John Carew Eccles AC Kt FAA FRS shared with Sir Alan Lloyd Hodgkin OM KBE FRS and Sir Andrew Fielding Huxley OM PRS ‘for their discoveries concerning the ionic mechanisms involved in excitation and inhibition in the peripheral and central portions of the nerve cell membrane’.
1960 Nobel Prize in Physiology or Medicine
Sir (Frank) Macfarlane Burnet OM AK KBE FAA FRS shared with Sir Peter Brian Medawar OM CH FRS ‘for discovery of acquired immunological tolerance’.
1945 Nobel Prize in Physiology or Medicine
Sir Howard Walter Florey, later Baron Florey of Adelaide and Marston OM FRS, shared with Sir Ernst Boris Chain FRS and Sir Alexander Fleming FRS ‘for the discovery of penicillin and its curative effect in various infectious diseases’.
1915 Nobel Prize for Physics
Sir (William) Lawrence Bragg CH OBE MC FRS and Sir William Henry Bragg OM KBE FRS ‘for their services in the analysis of crystal structure by means of X-rays’.
Historical Records of Australian Science


Historical Records of Australian Science documents the history of science, pure and applied, in Australia and the southwest Pacific. It also features biographical memoirs of deceased Fellows of the Australian Academy of Science commissioned by the Council of the Academy, and includes an annual bibliography of the history of Australian science.
The journal began in 1966 as an irregular publication under the title Records of the Australian Academy of Science, the present name being adopted in 1980. Since then, the journal has appeared on a regular basis, at first annually and, since 1991, twice a year.
Published by CSIRO Publishing on behalf of the Academy, the journal offers open access options.
Visit Historical Records of Australian Science | ConnectSci to read the latest articles and memoirs in Historical Records of Australian Science and sign up to the journal's email alert to receive the latest content straight to your inbox.
Explore our collection

One of the great changes in the archive and library world in the twenty-first century is not taking place in collection or gallery spaces. Instead, this transformation is taking place online. The Academy is making an ever-increasing number of collection resources available in digital form, but there is still much to be done and so we are open to the public by appointment to visit the physical collections in Canberra.
Contact the Academy to enquire.
Cultural Warning
Aboriginal and Torres Strait Islander people are advised that the Basser Library and Fenner Archives contains images, sounds and names of deceased persons. The Academy wishes to acknowledge that some material in the collections may be considered culturally sensitive.
Terms and annotations that reflect the author’s view, or those of the time in which an item was created, may not be considered appropriate today. These views are not endorsed by the Academy but are retained in their original form in an historical context.







