Sectional Committees

SC1 - Mathematics
- Professor Aurore Delaigle FAA | Chair
- Professor Andrew Hassell FAA | Member
- Professor Noel Cressie FAA | Member
- Professor Kate Smith-Miles AO FAA | Member
- Professor Jessica Purcell FAA | Member
SC2 - Physics and Astronomy
- Professor Joss Bland-Hawthorn FAA | Chair
- Professor Christine Charles FAA | Member
- Professor Igor Bray AM FAA | Member
- Professor Mark Krumholz FAA | Member
- Professor Margaret Reid FAA | Member
SC3—Chemistry
- Professor Frank Caruso FAA FTSE FRS | Chair
- Professor Maria Forsyth AM FAA | Member
- Professor Dmitri Golberg FAA | Member
- Emerita Professor Mary Garson AM FAA | Member
- Professor Elaine Holmes FAA | Member
- Professor Rich Payne FAA | Member
SC4—Earth and Planetary Sciences
- Professor Hugh O'Neill FAA FRS | Chair
- Professor Nerilie Abram FAA | Member
- Professor Hrvoje Tkalčić FAA | Member
- Professor Craig Simmons FAA FTSE | Member
- Emeritus Professor Greg Houseman FRS | Member
- Emeritus Professor Zheng-Xiang Li FAA | Member
SC5—Engineering sciences
- Professor Liming Dai FAA FTSE | Chair
- Professor John Carter AM FAA FTSE | Member
- Professor Yuri Estrin FAA | Member
- Dr Anita Hill FAA FTSE | Member
- Professor Tony Weiss AM FAA FTSE FAHMS | Member
- Professor Xiaojing Hao FAA FTSE | Member
SC6—Information and communication sciences
- Professor Albert Zomaya FAA | Chair
- Professor Ian Petersen FAA | Member
- Dr Greg Clark AC FAA FTSE | Member
- Professor Xinghuo Yu FAA | Member
- Emeritus Professor Rick Middleton FAA | Member
- Professor Rob Evans FAA FTSE | Member
SC7—Plant and animal sciences
- Professor Leslie Weston FAA | Chair
- Dr Jean Finnegan FAA | Member
- Professor Anna Koltunow FAA FTSE | Member
- Professor Robin Gasser FAA | Member
- Professor Ute Roessner AM FAA | Member
SC8 - Ecology, environment and evolution
- Professor Hugh Possingham FAA FRS | Chair
- Professor Mark Burgman FAA | Member
- Professor Linda Blackall FAA | Member
- Professor David Keith FAA | Member
- Professor Belinda Medlyn FAA | Member
- Professor Nicole Webster FAA | Member
SC9—Molecular and cell biology, and human genetics
- Professor Susan Clark FAA FAHMS | Chair
- Professor Rob Parton FAA | Member
- Professor Chris Mitchell AO FAA FAHMS | Member
- Professor Jozef Gécz FAA FAHMS | Member
- Professor David Komander FAA FRS | Member
SC10—Immunology, microbiology and haematology
- Professor Mariapia Degli-Esposti FAA FAHMS | Chair
- Professor Stephen Nutt FAA | Member
- Professor Gabrielle Belz FAA FAHMS | Member
- Professor Jian Li FAA | Member
- Professor Jeff Errington FAA FRS | Member
SC11—Physiology and neuroscience
- Professor Sam Berkovic AC FAA FAHMS FRS | Chair
- Professor David Gardner AM FAA | Member
- Professor Lois Salamonsen FAA | Member
- Professor Matthew Kiernan AM FAA FAHMS | Member
- Professor Pankaj Sah FAA FAHMS | Member
SC12—Medicine, dentistry and health sciences
- Professor Arthur Christopoulos FAA FAHMS | Chair
- Professor Patrick McGorryAO FAA FAHMS | Member
- Professor Michelle Haber AM FAA FAHMS | Member
- Professor Andrew Wilks FAA FTSE FAHMS | Member
- Professor Helen Christensen AO FAA FAHMS FASSA | Member
SC13—Interdisciplinary
- Professor Geoff McLachlan FAA | Chair
- Dr Jenny Stauber FAA FTSE | Member
- Professor Budiman Minasny FAA | Member
- Professor David Burke AC FAA FTSE | Member
Corresponding Member and Special Election Committee
- Professor John Shine AC FAA FAHMS(Hon) FRS | Chair
- Professor Marilyn Anderson AO FAA FTSE | Member
- Professor Helene Marsh AO FAA FTSE | Member
- Professor Colin Raston AO FAA | Member
Election to the Academy


The Australian Academy of Science aims to be a leader in diversity and inclusion in Australia’s science sector. We are committed to supporting excellence in science, but we recognise that to achieve this we must celebrate and embrace diversity and inclusion in all its forms.
More about the Academy’s approach to diversity and inclusion
Watch our videos encouraging the nominations of diversity candidates
As only our Fellows can nominate candidates for election to the Academy, we encourage all our Fellows to nominate candidates from diverse backgrounds from across the nation.
To assist us to identify Fellowship candidates from a broad range of backgrounds, each year we invite over 300 leaders in the Australian STEM and tertiary sectors to suggest candidates to us by mid-June. The Academy’s Champions of Diversity (Fellows appointed in each area of science) assess the suggested candidates for their suitability for election, and an appropriate Fellow is then identified to nominate the candidate. This involves working closely with the candidate on the nomination, which the proposer must start by 31 July in our online nomination system, with the full nomination submitted by 31 August.
Each year the Academy may elect up to 24 new Fellows by ‘Ordinary Election’ and up to four additional Fellows by ‘Special Election’. In June 2025, the Fellowship comprised 634 Fellows, including 132 women.
The Academy’s Council may also elect up to two Corresponding Members each year from among distinguished scientists overseas. As at June 2025 there were 39 Corresponding Members in the Academy.
Candidates may be considered for up to 5 election rounds (or until they are elected). The next round is the 2026 election round (which opened July 1 2025 with the new Fellows announced in May 2026).
Timeline: 2026 election round
| Date | Activity |
|---|---|
| By Mid-June | Candidates from diverse backgrounds may be suggested (in-confidence) to the Fellowship Officer by email to fellowship@science.org.au. Suggested candidates who are assessed by the Academy’s Champions of Diversity as suitable for election will then have an appropriate Fellow identified to nominate the candidate. |
| 1 July | Online nomination system opens for Fellows to nominate new candidates (and to start updating nominations for their candidates who will be considered again in this round). |
| By 31 July | All new nominations must be registered by 31 July. To register a candidate, start a new nomination in the system and enter and save the candidate’s name, email, gender, and the proposed Sectional Committee to assess the candidate. |
| By 31 August | All nominations must be completed and submitted by 31 August. This includes new nominations and updates to continuing nominations. |
| September to November | 13 selection committees, known as ‘Sectional Committees’, undertake unconscious bias training and then shortlist candidates within their discipline area/s to progress for further consideration. Additional independent referee reports are requested for shortlisted candidates. |
| By early February 2026 | Sectional Committees conduct their final deliberations to determine up to four candidates to recommend to Council. |
| March 2026 | Council considers all the recommended candidates and determines the final list of up to 24 candidates for the Fellows’ ballot for Ordinary Election, and up to 4 candidates for Special election. |
| April 2026 | Ballot count by Returning Officer and new Fellows declared elected (all Fellows advised under embargo). Two-thirds of the voting Fellows must agree to each candidate’s election. |
| End of May 2026 | New Fellows and Corresponding Members publicly announced. |

Professor Kerrie Mengersen FAA, QUT SEF. Credit: Vanessa Hunter
Eligibility for election
- Candidates for Ordinary and Special Election must be Australian citizens or permanent residents in Australia, and have an attachment to an Australian research organisation. For recent arrivals to Australia, who are not Australian citizens, candidates should be a resident for the two years preceding the date of the election meeting in early February and contributing to Australian science.
- Candidates for Corresponding Membership shall be persons, not normally resident in Australia, who are eminent in some branch of natural knowledge.
Criteria for Ordinary Election
- Scientific achievement: (This dimension focuses on the candidate’s research including the translation or applied impact of that research). Scientific excellence is based on contributions with major impact in the field. This may be either a single contribution or multiple contributions with clear impact. (60-85% weighting). Indicators of impact may include but are not limited to: main publications; citation of those publications; ‘textbook’ science; patents; significant impact on practice or translation of the research; or other indicators relative to the standards for each discipline. For ‘applied’ candidates (where there is a significant impact of their applications of scientific knowledge to the invention or development of new devices, constructions, products or processes, or to the advancement of human health, welfare or the environment) these additional indicators of impact should also be addressed.
- National and international profile: (This dimension focuses on how others have responded to the candidate’s collective achievements). (10-35% weighting).
Indicators include: invitations to speak; grants and Fellowships; journal editorships; honours, awards, and prizes; membership of prestigious organisations or committees; and supportive letters from referees. - Leadership, mentorship, promotion of science and potential to advance the work of the Academy: (This dimension focuses on what the candidate has done other than their own research). (5-30% weighting).
Indicators include: executive and leadership roles in science (not just within the specific research area of the candidate); conference organisation; high-level peer review functions; science education, outreach, advocacy and/or policy development; successful mentorship of the next generation of scientists; other contributions to the discipline.
Since the 2024 Round, more flexibility in the weightings of the criteria for election have been adopted to enable Sectional Committees and Council to better assess contributions of candidates who may not have had a full-time research career path or opportunities to undertake leadership roles.
The weighting of criterion 1 is a minimum of 60% and a maximum of 85%, however, criteria 2 and 3 may also now be adjusted by the Sectional Committee (to range from 15 to 40% combined) with a minimum weighting of 10% for criteria 2 and 5% for criteria 3.
Proposers are not to choose weightings. This information is provided for reference only. The flexibility is for Sectional Committees to adjust the weightings as they see fit given the circumstances as described in the nomination. Normally the weighting used would be: 60:20:20. These changes are only to allow full appreciation of non-traditional career pathways, and any changes used by a committee need to be commented on in the Chair’s report to Council.

The Academy is committed to celebrating and supporting diversity within the Fellowship. Achievement is judged relative to opportunity, taking into account any breaks in, or late commencement of, career. Assessment is done in such a way as to not disadvantage gifted young scientists who have already made outstanding contributions to their discipline. Diversity dimensions within the Fellowship (including gender, age, culture, state and region of residence, emerging disciplines, and interdisciplinary science) will be further taken into-account by Council in considering the final list of candidates.
Criteria for Special Election
Each year the Council may elect up to four persons whose election would expand the diversity of the Fellowship and be of significant benefit to the Academy and to the advancement of science. It is expected that candidates for Special Election will be scientists, engineers, or inventors and have a high national and/or international profile and potential to advance the work of the Academy in science education, advocacy, policy, or international engagement. They may also have a strong record of science achievement, usually early in their career. Specially elected Fellows will have:
- rendered conspicuous service to the cause of science, OR
- demonstrated outstanding innovation or entrepreneurial spirit through the translation of the results of scientific research that have led to global impact.
Special election candidates will be assessed using the following indicators of merit:
- Evidence of sustained service to the cause of science and science leadership, including mentorship, at the highest level and/or evidence of innovation or entrepreneurial spirit through the translation of the results of scientific research that have led to global impact. (50%)
- Evidence of very high national and/or international profile, including supportive letters of reference from eminent referees. (30%)
- Potential to advance the work of the Academy in areas of strategic importance to the Academy in science education, advocacy, policy, or international engagement. (20%)
Criteria for Corresponding Membership
- The primary selection criterion is scientific excellence through EITHER a single contribution to science, OR a cohesive body of smaller contributions with clear impact, as indicated by but not limited to: significant publications, citation of those publications, ‘textbook’ science, patents, major impact on practice or translation of the research, or other indicators relative to the standards for each discipline. (50%)
- Extremely high international research profile, including supportive letters of reference from eminent referees. (30%)
- Strength of connection or strategic value to Australian science including potential to advance the work of the Academy in science education, advocacy or policy. (20%)
Nomination for Fellowship

Professor Geordie Williamson FAA FRS
Fellows nominate candidates via the Academy’s nomination system. Information required includes:
- signed agreement from the candidate to nomination
- candidate declaration of research integrity
- proposer declaration regarding the research integrity of the candidate
- short citation
- support from at least four Fellows
- extended citation against the selection criteria
- full curriculum vitae
- list of 10 significant publications that have changed the field, with description demonstrating ‘impact in the field’ (rather than the 10 highest cited). If significant impact is across more than one field and the candidate is being considered by more than one Sectional Committee (a primary and a secondary committee) or by the Interdisciplinary Committee, then more than 10 papers (up to 20) would be allowable
- full publication list
- referees willing to support the nomination
A nomination is valid for five consecutive election rounds/years or until the candidate is elected. Proposers must update the nominations for their continuing candidates every year for the life of the nomination. After the nomination has expired and following a pause of two election rounds (two years), a new nomination for that candidate may be submitted.
Please email fellowship@science.org.au if you have any questions regarding the nomination process (including the eligibility of a candidate or suitability of a referee). Enquiries will be referred to the Secretary for Physical Sciences or the Secretary for Biological Sciences, where necessary.
You are welcome to share and embed our videos encouraging the nomination of diversity candidates for Fellowship and awards.
Genetic modification

Do you have questions about genetic modification?
The Australian Academy of Science strives to ensure the Australian community is guided by and enjoys the benefits of scientific endeavour. The purpose of this booklet is to provide a clear, concise description of genetic modification technology for Australians, including how it is used and how it is regulated. The booklet draws on the knowledge and expertise of the Academy’s distinguished Fellows and of the Australian science community, to provide information about this important scientific issue.
The booklet was prepared by an expert working group and reviewed by an oversight committee chaired by Professor John Shine AO PresAA, President of the Australian Academy of Science.
Download the PDF
Genetic modification booklet (PDF 1.2 MB, 20 pages)
Immunisation references

- Plotkin, S. A. et al. The science of vaccine safety: Summary of meeting at Wellcome Trust. in Vaccine vol. 38 1869–1880 (Elsevier Ltd, 2020).
- Plotkin, S., Orenstein, W. & Offit, P. Vaccines. The Lancet Infectious Diseases vol. 8 (Elsevier, 2008).
- Roush, S. W. et al. Historical comparisons of morbidity and mortality for vaccine-preventable diseases in the United States. Journal of the American Medical Association vol. 298 2155–2163 (2007).
- Simons, E. et al. Assessment of the 2010 global measles mortality reduction goal: Results from a model of surveillance data. Lancet 379, 2173–2178 (2012).
- ASCIA Acute Management of Anaphylaxis. www.allergy.org.au/hp/papers/acute-management-of-anaphylaxis-in-pregnancy (2020).
- Pollard, A. J. & Bijker, E. M. A guide to vaccinology: from basic principles to new developments. Nature Reviews Immunology vol. 21 83–100 (2020).
- Lemon, S. M. & Thomas, D. L. Vaccines to Prevent Viral Hepatitis. N. Engl. J. Med. 336, 196–204 (1997).
- Neuzil, K. M., Dupont, W. D., Wright, P. F. & Edwards, K. M. Efficacy of inactivated and cold-adapted vaccines against influenza A infection, 1985 to 1990: The pediatric experience. Pediatr. Infect. Dis. J. 20, 733–740 (2001).
- Stratton, K., Ford, A., Rusch, E. & Clayton, E. W. Adverse effects of vaccines: Evidence and causality. Adverse Effects of Vaccines: Evidence and Causality (National Academies Press, 2012). doi:10.17226/13164.
- Barlow, W. E. et al. The Risk of Seizures after Receipt of Whole-Cell Pertussis or Measles, Mumps, and Rubella Vaccine. N. Engl. J. Med. 345, 656–661 (2001).
- Mantadakis, E., Farmaki, E. & Buchanan, G. R. Thrombocytopenic Purpura after Measles-Mumps-Rubella Vaccination: A Systematic Review of the Literature and Guidance for Management. J. Pediatr. 156, 623–628 (2010).
- Perry, R. T. & Halsey, N. A. The clinical significance of measles: A review. Journal of Infectious Diseases vol. 189 (2004).
- Pulendran, B. & Ahmed, R. Immunological mechanisms of vaccination. Nature Immunology vol. 12 509–517 (2011).
- Stowe, J., Andrews, N., Wise, L. & Miller, E. Investigation of the temporal association of Guillain-Barré syndrome with influenza vaccine and influenzalike illness using the United Kingdom general practice research database. Am. J. Epidemiol. 169, 382–388 (2009).
- Wraith, D. C., Goldman, M. & Lambert, P. H. Vaccination and autoimmune disease: What is the evidence? Lancet vol. 362 1659–1666 (2003).
- Alberts, B. et al. Molecular biology of the cell. Biochemistry and Molecular Biology Education vol. 36 (Wiley, 2008).
- Murphy, K. M., Travers, P. & Walport, M. Janeway’s Immunobiology. (Garland Science, 2007).
- Banchereau, J. & Steinman, R. M. Dendritic cells and the control of immunity. Nature vol. 392 245–252 (1998).
- Miller, J. F. Immunological function of the thymus. Lancet 278, 748–749 (1961).
- Miller, J. F. & Mitchell, G. F. Cell to cell interaction in the immune response. I. Hemolysin-forming cells in neonatally thymectomized mice reconstituted with thymus or thoracic duct lymphocytes. J. Exp. Med. 128, 801–820 (1968).
- Roitt, I. M., Greaves, M. F., Torrigiani, G., Brostoff, J. & Playfair, J. H. The cellular basis of immunological responses. A synthesis of some current views. Lancet vol. 2 367–371 (1969).
- Zinkernagel, R. M. & Doherty, P. C. Restriction of in vitro T cell-mediated cytotoxicity in lymphocytic choriomeningitis within a syngeneic or semiallogeneic system. Nature 248, 701–702 (1974).
- Burnet, F. M. The Clonal Selection Theory of Acquired Immunity. (Cambridge University Press, 1959).
- Ahmed, R. & Gray, D. Immunological memory and protective immunity: Understanding their relation. Science (80-. ). 272, 54–60 (1996).
- Sallusto, F., Lanzavecchia, A., Araki, K. & Ahmed, R. From vaccines to memory and back. Immunity vol. 33 451–463 (2010).
- Panum, L. P. Observations made during the epidemic of measles on the Faroe Islands in the year 1846. Bibiliothek for Laeger 3R, 270–344 (1847).
- Adkins, B., Leclerc, C. & Marshall-Clarke, S. Neonatal adaptive immunity comes of age. Nature Reviews Immunology vol. 4 553–564 (2004).
- Chen, R. T. et al. Measles Antibody: Reevaluation of Protective Titers. J. Infect. Dis. 162, 1036–1042 (1990).
- Schauer, U. et al. Levels of antibodies specific to tetanus toxoid, Haemophilus influenzae type b, and pneumococcal capsular polysaccharide in healthy children and adults. Clin. Diagn. Lab. Immunol. 10, 202–207 (2003).
- Weibel, R. E., Stokes, J., Buynak, E. B., Whitman, J. E. & Hilleman, M. R. Live, Attenuated Mumps-Virus Vaccine. N. Engl. J. Med. 276, 245–251 (1967).
- Offit, P. A. & Jew, R. K. Addressing Parents’ Concerns: Do Vaccines Contain Harmful Preservatives, Adjuvants, Additives, or Residuals? Pediatrics vol. 112 1394–1401 (2003).
- Stone, C. A. et al. Anaphylaxis after zoster vaccine: Implicating alpha-gal allergy as a possible mechanism. J. Allergy Clin. Immunol. 139, 1710-1713.e2 (2017).
- Stone, C. A. et al. Anaphylaxis after vaccination in a pediatric patient: further implicating alpha-gal allergy. J. Allergy Clin. Immunol. Pract. 7, 322-324.e2 (2019).
- Delrue, I., Verzele, D., Madder, A. & Nauwynck, H. J. Inactivated virus vaccines from chemistry to prophylaxis: Merits, risks and challenges. Expert Review of Vaccines vol. 11 695–719 (2012).
- Dull, P. M. & McIntosh, E. D. Meningococcal vaccine development - from glycoconjugates against MenACWY to proteins against MenB - potential for broad protection against meningococcal disease. Vaccine 30, (2012).
- Parkman, P. D. & Hopps, H. E. Viral vaccines and antivirals: Current use and future prospects. Annual Review of Public Health vol. 9 203–221 (1988).
- Zuckerman, A. J. Subunit, recombinant and synthetic hepatitis b vaccines. Scand. J. Gastroenterol. 20, 27–38 (1985).
- Avci, F. Y., Li, X., Tsuji, M. & Kasper, D. L. A mechanism for glycoconjugate vaccine activation of the adaptive immune system and its implications for vaccine design. Nat. Med. 17, 1602–1609 (2011).
- Lauring, A. S., Jones, J. O. & Andino, R. Rationalizing the development of live attenuated virus vaccines. Nature Biotechnology vol. 28 573–579 (2010).
- Edelman, R. Vaccine adjuvants. Reviews of infectious diseases vol. 2 370–383 (1980).
- Einstein, M. H. et al. Comparison of the immunogenicity and safety of CervarixTM and Gardasil® human papillomavirus (HPV) cervical cancer vaccines in healthy women aged 18-45 years. Hum. Vaccin. 5, 705–719 (2009).
- Dey, A. et al. Surveillance of adverse events following immunisation in Australia: annual report, 2018. Commun. Dis. Intell. 44, (2020).
- McNeil, M. M. et al. Risk of anaphylaxis after vaccination in children and adults. J. Allergy Clin. Immunol. 137, 868–878 (2016).
- Imafidon, G. I. & Sosulski, F. W. Nucleic Acid Nitrogen of Animal and Plant Foods. J. Agric. Food Chem. 38, 118–120 (1990).
- Wilson, D., Minor, P. D., Afzal, M. A. & Ghosh, S. Detection of persistent measles virus infection in Crohn’s disease: Current status of experimental work. Gut vol. 48 748–752 (2001).
- Khakoo, G. A. & Lack, G. Recommendations for using MMR vaccine in children allergic to eggs. BMJ Br. Med. J. 320, 929 (2000).
- Mullins, R. J., Kemp, A. & Gold, M. Influenza vaccination of the egg-allergic individual. Medical Journal of Australia vol. 193 254–255 (2010).
- Davidkin, I., Kontio, M., Paunio, M. & Peltola, H. MMR vaccination and disease elimination: The finnish experience. Expert Review of Vaccines vol. 9 1045–1053 (2010).
- Campbell, H., Andrews, N., Brown, K. E. & Miller, E. Review of the effect of measles vaccination on the epidemiology of SSPE. International Journal of Epidemiology vol. 36 1334–1348 (2007).
- Nebbia, G., Peppa, D. & Maini, M. K. Hepatitis B infection: Current concepts and future challenges. QJM vol. 105 109–113 (2012).
- Pol, S., Vallet-Pichard, A., Corouge, M. & Mallet, V. O. Hepatitis C: Epidemiology, diagnosis, natural history and therapy. Contributions to Nephrology vol. 176 1–9 (2012).
- El-Serag, H. B. Hepatocellular Carcinoma. N. Engl. J. Med. 365, 1118–1127 (2011).
- McLaughlin-Drubin, M. E., Meyers, J. & Munger, K. Cancer associated human papillomaviruses. Current Opinion in Virology vol. 2 459–466 (2012).
- Schiffman, M., Castle, P. E., Jeronimo, J., Rodriguez, A. C. & Wacholder, S. Human papillomavirus and cervical cancer. Lancet vol. 370 890–907 (2007).
- Kimberlin, D. W. & Whitley, R. J. Varicella–Zoster Vaccine for the Prevention of Herpes Zoster. N. Engl. J. Med. 356, 1338–1343 (2007).
- Kim, T. H., Johnstone, J. & Loeb, M. Vaccine herd effect. Scandinavian Journal of Infectious Diseases vol. 43 683–689 (2011).
- Simon, A. K., Hollander, G. A. & McMichael, A. Evolution of the immune system in humans from infancy to old age. Proceedings of the Royal Society B: Biological Sciences vol. 282 (2015).
- Kahn, C. R. Perverted priorities. The physician/scientist as spokesman and salesman for biomedical research. Presidential address to the American Society for Clinical Investigation, Washington, DC, 29 April 1989. The Journal of clinical investigation vol. 84 723–727 (1989).
- World Health Organisation. Hepatitis B vaccines. https://www.who.int/immunization/HepB_position_paper_oct09_summary.pdf (2009).
- Petrova, V. N. et al. Incomplete genetic reconstitution of B cell pools contributes to prolonged immunosuppression after measles. Sci. Immunol. 4, 6125 (2019).
- Mina, M. J. et al. Measles virus infection diminishes preexisting antibodies that offer protection from other pathogens. Science (80-. ). 366, 599–606 (2019).
- Beyer, W. E. P., Palache, A. M., Kerstens, R. & Masurel, N. Gender differences in local and systemic reactions to inactivated influenza vaccine, established by a meta-analysis of fourteen independent studies. Eur. J. Clin. Microbiol. Infect. Dis. 15, 65–70 (1996).
- Peltola, H. & Heinonen, O. Frequency of true adverse reactions to measles-mumps-rubella vaccine. A double-blind placebo-controlled trial in twins. Lancet 327, 939–942 (1986).
- Klein, N. P. et al. Measles-mumps-rubella-varicella combination vaccine and the risk of febrile seizures. Pediatrics 126, (2010).
- Parker, S. K., Schwartz, B., Todd, J. & Pickering, L. K. Thimerosal-containing vaccines and autistic spectrum disorder: A critical review of published original data. Pediatrics 114, 793–804 (2004).
- Poehling, K. A. et al. The Underrecognized Burden of Influenza in Young Children. N. Engl. J. Med. 355, 31–40 (2006).
- White, O. J. et al. A genomics-based approach to assessment of vaccine safety and immunogenicity in children. Vaccine 30, 1865–1874 (2012).
- Andre, F. et al. Vaccination greatly reduces disease, disability, death and inequity worldwide. WHO (2011).
- Rappuoli, R., Mandl, C. W., Black, S. & De Gregorio, E. Vaccines for the twenty-first century society. Nature Reviews Immunology vol. 11 865–872 (2011).
- Monto, A. S. & Petrie, J. G. Improving Influenza Vaccine Effectiveness: Ways to Begin Solving the Problem. Clinical Infectious Diseases vol. 69 1824–1826 (2019).
- Li, N., Peng, L. H., Chen, X., Nakagawa, S. & Gao, J. Q. Transcutaneous vaccines: Novel advances in technology and delivery for overcoming the barriers. Vaccine vol. 29 6179–6190 (2011).
- Van Der Maaden, K., Jiskoot, W. & Bouwstra, J. Microneedle technologies for (trans)dermal drug and vaccine delivery. Journal of Controlled Release vol. 161 645–655 (2012).
- Mbow, M. L., De Gregorio, E., Valiante, N. M. & Rappuoli, R. New adjuvants for human vaccines. Current Opinion in Immunology vol. 22 411–416 (2010).
- Rybicki, E. P. Plant-produced vaccines: promise and reality. Drug Discovery Today vol. 14 16–24 (2009).
- Garland, S. M. et al. Noninferiority of antibody response to human papillomavirus type 16 in subjects vaccinated with monovalent and quadrivalent L1 virus-like particle vaccines. Clin. Vaccine Immunol. 14, 792–795 (2007).
- Weibel, R. E., Buynak, E. B., McLean, A. A. & Hilleman, M. R. Long-Term Follow-up for Immunity After Monovalent or Combined Live Measles, Mumps, and Rubella Virus Vaccines. Pediatrics 56, (1975).
- Rappuoli, R. & Aderem, A. A 2020 vision for vaccines against HIV, tuberculosis and malaria. Nature vol. 473 463–469 (2011).
- Rupnow, M. F. T., Chang, A. H., Shachter, R. D., Owens, D. K. & Parsonnet, J. Cost-effectiveness of a potential prophylactic helicobacter pylori vaccine in the united states. J. Infect. Dis. 200, 1311–1317 (2009).
- Howie, S. E. M., Horner, P. J., Horne, A. W. & Entrican, G. Immunity and vaccines against sexually transmitted Chlamydia trachomatis infection. Curr. Opin. Infect. Dis. 24, 56–61 (2011).
- Pardi, N., Hogan, M. J., Porter, F. W. & Weissman, D. mRNA vaccines-a new era in vaccinology. Nature Reviews Drug Discovery vol. 17 261–279 (2018).
- Nossal, G. J. V. Vaccines of the future. Vaccine 29, (2011).
- Cucinotta, D. & Vanelli, M. WHO declares COVID-19 a pandemic. Acta Biomedica vol. 91 157–160 (2020).
- Castells, M. C. & Phillips, E. J. Maintaining Safety with SARS-CoV-2 Vaccines. N. Engl. J. Med. 384, 643–649 (2021).
- Petousis-Harris, H. & Alley, L. Impact of antivaccination campaigns on health worldwide: lessons for Australia and the global community. Med. J. Aust. 213, 300-301.e1 (2020).
- Zhang, E. J., Chughtai, A. A., Heywood, A. & MacIntyre, C. R. Influence of political and medical leaders on parental perception of vaccination: A cross-sectional survey in Australia. BMJ Open 9, e025866 (2019).
- Leask, J. et al. Communicating with parents about vaccination: a framework for health professionals. BMC Pediatrics vol. 12 154 (2012).
- Brewer, N. T., Chapman, G. B., Rothman, A. J., Leask, J. & Kempe, A. Increasing Vaccination: Putting Psychological Science Into Action. Psychol. Sci. Public Interes. 18, 149–207 (2017).
The science of immunisation: About

Note from the President

Professor John Shine
The Australian Academy of Science is an independent institution that provides science advice and builds public awareness and understanding of science. This guide was created to help Australians understand scientific information about immunisation so that everyone can make well-informed healthcare decisions. It can be used as a helpful tool for discussions between healthcare professionals and patients, between family and friends, and any conversations about vaccination.
The Academy is grateful for the pro-bono contributions made to this report by the expert working group. The Academy also thanks the Australian Government Department of Health for providing financial support to prepare and publish this latest edition and previous versions of this guide.
Professor John Shine AC PresAA FRS
President, Australian Academy of Science
This edition of The Science of Immunisation (2021)
Expert Working Group
- Professor Carola Vinuesa FAA FAHMS (Chair)
Co-Director, Centre for Personalised Immunology, John Curtin School of Medical Research, Australian National University - Dr Preeya Alexander
General Practitioner and Medical Educator - Professor Tony Basten AO FAA FTSE
Emeritus Fellow, Garvan Institute of Medical Research - Dr Frank Beard
Associate Director, National Centre for Immunisation Research and Surveillance; Senior Lecturer, School of Public Health, University of Sydney - Professor Tony Cunningham AO FAHMS
Director, Centre for Virus Research, The Westmead Institute for Medical Research; Professor, Faculty of Medicine and Health, University of Sydney - Associate Professor Margie Danchin
Murdoch Children's Research Institute, The Royal Children's Hospital and University of Melbourne - Dr Antonio Di Dio
ACT President, Australian Medical Association - Mr Darius Everett
Acting Assistant Secretary, Immunisation and Communicable Disease Branch, Australian Government Department of Health - Dr Rod Lamberts
Deputy Director, Australian National Centre for the Public Awareness of Science, Australian National University - Professor Kristine Macartney
Director, National Centre for Immunisation Research and Surveillance; Professor in Paediatrics and Child Health, University of Sydney - Distinguished Professor Ingrid Piller FAHA
Professor of Applied Linguistics, Macquarie University - Ms Lisa Ranson
COVID Communications, Australian Government Department of Health
Peer Reviewers
- Professor Ian Frazer AC FAA FTSE FAHMS FRS
- Professor Dale Godfrey FAHMS
- Professor Patrick Holt FAA
- Professor Julie Leask
Acknowledgements
Project management, science policy, editorial services, design and communication assistance was provided by the Australian Academy of Science. Contributing staff members Dr Hayley Teasdale, Ms Emma Berthold, Mr Chris Anderson, Mr Andrew Berthold, Dr Tom Carruthers, Ms Robyn Diamond, Mr Paul Richards, Mr Dan Wheelahan and Ms Anna-Maria Arabia are gratefully acknowledged.
Previous editions
Expert Working Group
- Professor Tony Basten AO FAA FTSE (Co-chair)
- Professor Ian Frazer AC FAA FTSE FAHMS FRS (Co-chair)
- Professor Francis Carbone FAA
- Professor Patrick Holt FAA
- Professor Julie Leask
- Professor Peter McIntyre AO
- Professor Terry Nolan
- Professor Judith Whitworth AC
Reviewers
- Sir Gus Nossal AC CBE FAA FTSE FAHMS FRS (Chair)
- Professor Ian Gust AO
- Professor Fiona Stanley AC FAA FAHMS(Hon) FASSA
- Professor Bob Williamson AO FAA FRS FAHMS(Hon)
Copyright and ISBNs
The Australian Academy of Science received funding from the Australian Government Department of Health to publish this document.
Interest declarations of all committee members are available at www.science.org.au/disclosure-register
ISBN 978-0-85847-765-0 (Printed booklet)
ISBN 978-0-85847-766-7 (Digital PDF with references)
ISBN 978-0-85847-767-4 (Digital PDF without references)
© 2021 Australian Academy of Science
GPO Box 783, Canberra ACT 2601, all rights reserved. Selected passages, tables or diagrams may be reproduced, provided the source is acknowledged. Major extracts are not permitted without the written permission of the Academy.
Please cite “The science of immunisation: Questions and answers”, Australian Academy of Science, Canberra, 2021. www.science.org.au/immunisation
For a version with full references and figure sources, visit www.science.org.au/immunisation
Cover images (PDFs and printed versions): Unsplash and Kathie Teasdale
5. What does the future hold for vaccination?

Infectious diseases still account for around 40% of all recorded deaths in the world.
Advances in vaccine technology are crucial to limit and prevent infectious diseases around the world, which still account for around 40% of all recorded deaths globally.68 Changing how existing vaccines are used, developing new vaccine delivery technologies and generating new vaccines are some of the ways researchers are working to overcome this ongoing challenge and save lives.69
New technologies will improve vaccine delivery and efficacy
Many technologies under development will improve the effectiveness of vaccine delivery and make it simpler.70
To make a vaccine that only needs to be given once, it must either be very powerful or packaged so that its contents are released intermittently once it has been administered. Technologies and alternative adjuvants that can remove the need for multiple shots are under development.71–73
Currently, many vaccines need to be injected—an experience that people can find unpleasant, and may be a serious psychological barrier for some. Needle-free administration is already possible for some vaccines, such as live vaccines given orally (e.g. rotavirus). Researchers are working on edible plant-based vaccine materials, needle-free skin patches and microneedle injection technologies to get the vaccine through the skin without discomfort.71,72,74
Technologies for delivering multiple antigens in one injection are improving. Many different inactivated vaccines can already be given in one injection without impairing the immune response to any of them, and some live virus vaccines can also be given in one injection.75,76 That means fewer needles for patients and more efficient vaccine delivery overall.
New vaccines are needed for chronic and emerging infections
Most successful vaccines protect against acute (short-lived) infections largely through the production of antibodies. Vaccines for chronic (long-lasting) infections, especially for HIV, tuberculosis and malaria, remain a challenge. One of the primary reasons for this is that the viruses, bacteria and parasites causing these infections hide from the immune system inside the person’s cells. To overcome this, a different kind of immune response involving T cells is required instead of, or in addition to, an antibody response.77
There are some infections associated with serious long-term complications that we don’t yet have a vaccine for. For instance, infection with the bacterium Helicobacter pylori means patients are more likely to develop stomach cancer, and group A streptococcus infection is responsible for rheumatic fever, which is still a significant cause of death and disability in developing countries.78,79
Based on experience with emerging infections like Ebola and Zika, progress has been made in developing vaccines that use mRNA (a type of genetic material) to stimulate some of our own cells to temporarily produce antigens.80 The research and progress made in this field contributed to the speed of development for COVID-19 vaccines.
Use of vaccines for treatment as well as prevention of diseases
Vaccines also have the potential to be used to treat diseases, rather than prevent them. Such therapeutic vaccines are being targeted at persistent infections, such as shingles and those due to human papilloma virus. They are also being targeted at non-infectious conditions, including autoimmune disorders, tumours, allergies, and drug addiction.
In the case of tumours, the vaccine can either be directed against the tumour itself or be designed to amplify the anti-tumour immune response. For autoimmune or allergic disorders, vaccines are being designed to switch off unwanted immune responses (so-called ‘negative vaccination’), rather than switching on the useful immune response needed for infections and cancer.81
Promising trials are in progress for vaccines to treat nicotine and cocaine addictions.
The COVID-19 pandemic: a global challenge
COVID-19 infection is caused by the SARS-CoV-2 virus, which belongs to the coronavirus family. This family includes the original SARS-CoV virus responsible for the SARS epidemic of 2003 and several other coronaviruses that cause the common cold.
SARS-Cov-2 is thought to have been transmitted to humans by an animal carrier. The virus carries a ‘spike’ protein that allows it to attach to human body cells, where it then enters the cell and reproduces.
The World Health Organization officially declared COVID-19 a pandemic on 11 March 2020.82 To date, the disease has led to severe illness and death in millions of people around the world. COVID-19 mainly affects the lungs but can also damage other parts of the body, including blood vessels, kidneys, heart and brain.
The increased development speed of vaccines for COVID-19 are a result of improved and new vaccine technologies being available, substantial investment, international collaboration between scientists and increasing the speed of regulatory review.
The scale of the COVID-19 pandemic has demonstrated the essential role of vaccination in today’s world. Several vaccines were produced and implemented in vaccination programs globally within the first year after the onset of the pandemic, compared with the previous average vaccine development time of 10 years.83 This reflects advances in vaccine technology that will assist us with other infectious diseases, not just COVID-19.
The rapid development of vaccines for COVID-19 is also a result of substantial investment, international collaboration between scientists and increasing the speed of regulatory review. These factors, combined with advances in vaccine technology, have enabled effective vaccines to be created and brought to market in record time while completing clinical trial phases and receiving all mandated regulatory and safety approvals.
Vaccine uptake
COVID-19 vaccines are being heralded as one of the leading solutions to control the pandemic and resume our previous way of life. Ensuring public confidence in the COVID-19 vaccines is crucial to facilitate vaccine uptake.
Despite decades of scientific research into vaccine safety and effectiveness, some people may still have concerns about vaccines, or may think that some diseases are not severe enough to need a vaccine.84 The spread of misinformation about vaccines can also make it more difficult for people to understand whether a claim is based on credible scientific evidence.
Good communication about the risks of vaccine-preventable diseases and the safety of vaccines is important for everyone. Discussing concerns with a trusted health professional is one of the best ways for people to understand what scientists and public health professionals know about a disease and the best protection against it.85 However, it is equally important for health professionals to listen openly to those concerns.86
Ensuring that key groups are protected against infectious diseases is crucial. In Australia, between 30 and 50% of pregnant women currently receive maternal influenza vaccines, and while vaccination rates for children under five years are relatively high, there are still many more opportunities to ensure these groups are receiving the highest levels of protection. Making sure people are confident that vaccination is the best way to avoid diseases is important, especially in the case of a global pandemic like COVID-19.
Developing a better understanding of the practical barriers to vaccination, as well as the social, cognitive and emotional aspects of making decisions about vaccines, will help everyone to feel more confident about keeping their immunisations up to date.87 Although the science of vaccine development is critical, it can only be fully effective when people actually receive their vaccines. The act of vaccination saves lives.
Explore more
Watch the video
Read additional information
4. Are vaccines safe?

It is safer to be vaccinated than get the disease
People are more likely to experience a serious complication from a disease, rather than from the vaccine for that disease.
All vaccines in use in Australia provide benefits that far outweigh any risks.2–4,9–11,14,15
Benefits of vaccines include the prevention of disease, and reduced chances of getting sick, going to hospital or dying.2–4,9–15
Risks of vaccines may include minor and moderate side effects, and more rare serious side effects.
1 in 15 patients with diphtheria die from the disease, whereas serious side effects from the diphtheria vaccine are very rare.3
1 in 4 people chronically infected with hepatitis B will die from cirrhosis of the liver or from liver cancer. Vaccination can prevent chronic infection with hepatitis B.59
In the case of measles infection, the virus erases the memory cells for other infections like the flu and intestinal diseases.60 As a result, people can become susceptible again to catching these diseases despite being previously immunised against them.61
Many vaccines have the added advantage of generating more effective protection against pathogens than through naturally acquiring the infection. For example, a small amount of tetanus toxin can cause life-threatening disease but is not enough to generate enough protective antibodies to prevent disease if exposed again in the future. By contrast, the tetanus vaccine has enough of the inactivated toxoid in it to generate the level of protective antibodies needed to protect against serious illness.
Most reactions from vaccination are minor
A local reaction at the injection site, such as redness, is one sign the immune system is interacting with the vaccine. This is normal.
The great majority of side effects that follow vaccination are minor and short-lived.
The most common side effects for all vaccine types are ‘local’ reactions at the injection site, such as redness or swelling, which occur within hours and are caused by the vaccine. More general or ‘systemic’ reactions, such as fever or tiredness, can also occur after vaccination, but studies have shown that they are much less common than local reactions.62
Local reactions are signs that the vaccine is interacting with the immune system to generate a protective response. The nature of these reactions varies depending on the type of vaccine given.
For example, if a person develops a fever due to an inactivated vaccine, they almost always do so within 24–48 hours—the time when the immune system is immediately responding to the components of the vaccine. In contrast, the onset of fever caused by a live attenuated vaccine, such as the MMR vaccine, is delayed for 7–12 days. This is due to the time needed for the attenuated virus in the vaccine to multiply enough to produce a protective response from the immune system.63,64
Some adverse events coincide with, but are not caused by, vaccination
Symptoms such as fever, rashes, irritability and nasal snuffles are common, especially among children. Consequently, it can be difficult to determine how many of these reactions are caused by a vaccine when the ‘background rate’ (how often it occurs anyway) in the same age group is unknown.
In some cases, these kinds of reactions may be caused by the vaccine. But in other situations, the symptoms may be unrelated, occurring by chance at the same time as the vaccination. For this reason, scientists refer to these kinds of symptoms as ‘adverse events following immunisation’ to indicate that the vaccine may not cause events that follow vaccination.
Many adverse events following a vaccine injection are coincidental.
Current vaccines used in Australia have fewer side effects than previous vaccines.
Safety surveillance systems in countries like Australia require health care providers to report adverse events that occur following vaccination, regardless of the cause. The reports are compared with historical trends to identify any changes requiring special investigation and assess whether these adverse events are vaccine-related.
It can be misleading to rely on the reported numbers of adverse events. Several factors must be considered to determine if an event is coincidental or caused by the vaccine. Many adverse events are coincidental.
This study showed that many common symptoms that occur after a vaccine is given are not caused by the vaccine, but occur by chance at that same time.
Case study: adverse events following vaccination
Researchers in Finland analysed common symptoms in 581 pairs of twins after one twin received the MMR vaccine and the other was given a dummy vaccine made of sterile salty water (a placebo).63
1–6 days after the injection: the number of adverse events in the twin who received the MMR vaccine was almost identical to those in the twin who received placebo.
7–12 days after the injection: the vaccinated group had a measurable increase in symptoms that are known to be associated with receiving the attenuated measles vaccine, such as fever, irritability and rash. On the other hand, when researchers looked at the frequency of coughing or other cold-like symptoms—which are common symptoms at any time, regardless of vaccination—they found no difference between the two groups.
Some of the symptoms known to occur after MMR vaccine were also seen in the group who received the placebo, but at a lower rate.
In summary, this study showed that many common symptoms that occur after a vaccine is given are not caused by the vaccine, but occur by chance at that same time.63
The MMR vaccine does not cause autism
Medical conditions with unknown causes have been incorrectly linked to particular vaccines. The most prominent example over the past 25 years is the claimed link between the MMR vaccine and autism. Children with autism will often display the first clinical signs in their second year of life, which also happens to be the same time that the MMR vaccine is usually given.
The original suggestion that the MMR vaccine might be linked to autism was made in 1998. A research group proposed that the attenuated measles virus in the vaccine infected the intestine. The research group leader claimed this led to inflammation that resulted in lower absorption of nutrients needed for normal brain development, resulting in developmental conditions such as autism.
Many comprehensive studies subsequently ruled out this suggested link by showing conclusively that rates of autism are the same among children who have and have not been vaccinated. Ultimately, the original report was shown to be fraudulent and was retracted by the medical journal that published it.
Similarly, any link between autism and thiomersal, previously used in small quantities as a preservative in vaccines, has been ruled out.65
Febrile seizures
Febrile seizures are a convulsion in a child caused by a rise in their body temperature, usually a fever.
Serious side effects from vaccines are very rare
Potentially worrying side effects, such as febrile seizures, have been reported after vaccination. However, such side effects occur much less often with the vaccine than they would if a person caught the disease itself.2,10,12 This is well illustrated in young children by comparing the frequency of adverse events from the MMR vaccine with the frequency of adverse events with measles itself.10
1 in 10 young children develop a fever after receiving influenza vaccine8
9 in 10 children develop a fever after a proven influenza infection66
About 3 in every 10,000 children who receive the MMR vaccine develop a fever high enough to cause short-lived seizures. In contrast, the risk of such a fever is more than 30 times greater among children who develop the disease—affecting about 100 in 10,000 children. Importantly, measles vaccination has prevented an estimated 23.3 million deaths worldwide.
The frequency of side effects associated with some earlier vaccine preparations (no longer in use in developed countries such as Australia) was higher than with the current generation of vaccines.
Below: Likelihood of severe complications (among 1 million children aged under 5 years) from receiving the MMR vaccine compared to those from having measles2,10–12
| COMPLICATION | MMR VACCINE | MEASLES | |
|---|---|---|---|
| Uncommon complications | Seizures or convulsions brought on by fever | 300 children have seizures | 10,000 children have seizures or convulsions induced by fever |
| Rare complications | A temporary tendency to bruise or bleed more easily (thrombocytopenia) | 26 children develop thrombocytopenia | 330 children develop thrombocytopenia |
| Very rare complications | Severe allergic reactions (anaphylaxis) | Up to four children have a severe allergic reaction. This is readily treated with complete recovery | No anaphylaxis cases |
| Inflammation of the brain (encephalitis), which may result in permanent brain damage or death | A maximum of one child may develop encephalitis | 2000 children may develop encephalitis | |
| Subacute sclerosing panencephalitis (SSPE), which causes progressive brain damage and death | No children will get SSPE | 10 children get SSPE several years later |
Vaccines have not led to a rise in autoimmune diseases
Autoimmune disease
Autoimmune diseases occur when the immune system mistakenly attacks the body instead of invading pathogens.
Over the past 30 years, the number of people who develop autoimmune diseases has been increasing, particularly in societies where rates of infectious disease have declined. This has raised the question of whether vaccine use can lead to the development of autoimmune disorders. Except for the two rare diseases mentioned below, the answer is no. This conclusion is based on the stringent monitoring procedures put in place for detecting side effects of vaccination.
The first exception is the small increase in the risk of developing the rare condition known as immune thrombocytopenic purpura, a condition where blood fails to clot normally, after receiving the MMR vaccine. However, the risk of developing this disorder associated with measles infection itself is more than 10 times greater than that associated with the vaccine.
The other exception is Guillain–Barré syndrome, a nerve condition, following influenza vaccination. Again, the risk of developing the disease after the influenza vaccination is much lower than after influenza infection.
Vaccines do not cause allergic diseases
Like autoimmune diseases, allergic diseases such as asthma have become more common in the developed world over the past 30 years. However, there is no significant evidence that vaccines cause allergic diseases in otherwise healthy people.
Generally, for every 100,000 doses of a vaccine, less than 1 person will experience a severe allergic reaction after receiving it—a rate that is extremely low.43 Nevertheless, people with a history of reactions to a specific vaccine or vaccine additives, or a strong family history of allergic disease, should always take precautions.
Injectable vaccines used in Australia do not contain detectable amounts of antibiotics such as penicillin or sulphonamides to which some people may be allergic. The hepatitis B vaccine is grown in yeast. Although there have been some isolated reports of possible severe allergic reactions to this vaccine, the benefits of receiving the vaccine far outweigh the multiple risks associated with hepatitis B infection.
Safety testing is an essential component of vaccine development and use

Careful surveillance continues even after a vaccine candidate has proven to be effective and has passed all safety checks. Image: Self Magazine CC-BY-2.0
Careful testing of vaccine safety is an essential part of vaccine development and manufacture. There are also ongoing surveillance programs after vaccines have been introduced into the community.
Before a vaccine can be developed, research is undertaken to better understand the pathogen and the disease it causes, helping to determine how potential vaccines are likely to work.
During vaccine development, safety testing procedures occur in multiple stages:
The first stage involves preclinical assessment in the laboratory, usually using animals.67 If a vaccine fails these safety tests, it cannot progress into clinical trials.
Vaccines are then evaluated in three phases of clinical trials:
In Phase I clinical trials, the potential vaccine (or ‘vaccine candidate’) is given to small numbers (typically 25–50) of healthy adults with the primary goal of assessing safety.
Phase II clinical trials involve hundreds of participants and are designed to show how good a vaccine is in provoking an immune response and determining the best dosage to use.
Phase III clinical trials aim to demonstrate a vaccine’s safety and how well it protects against the target disease across different groups of people, which usually requires administering the vaccine to many thousands of potentially susceptible people. Only after the vaccine has passed each of these safety and efficacy hurdles is it approved for widespread community use.
Some side effects of vaccines are so rare that they are not detected during the extensive safety testing before a vaccine is approved for use. To ensure that even very rare side effects are detected, careful surveillance continues even after a vaccine candidate has proven to be effective and has passed all safety checks. The formal term for this collection of information and reporting of any suspected adverse events is post-licensure assessment.
Rapid response to risks associated with vaccines
If a potential problem with a vaccine is detected, the use of that vaccine may be temporarily paused. This allows health authorities to investigate what’s caused the problem and whether it can be linked to a particular batch of the vaccine. For example, young children given a particular influenza vaccine in Australia in 2010 showed an increased risk of febrile seizures. When the problem first became apparent, the use of all influenza vaccines in young children was suspended to allow authorities to identify the one type of vaccine preparation causing the problem. Then, influenza vaccines shown not to be associated with unacceptable rates of febrile seizures were reintroduced to ensure that protection against influenza remained available for children at high risk of complications from the disease.
Explore more
Watch the video
Read additional information
3. Who benefits from vaccines?

Individuals benefit, in the short and long term
Efficacy
Efficacy refers to how well a vaccine performs in preventing new infections as it goes through scientific testing.
An effective vaccine protects an individual against a specific infectious disease and its complications. In the short term, vaccine efficacy is measured by its ability to reduce new infections. The longer-term goal is to reduce serious complications and death.2
All vaccines currently used in Australia produce high levels of protection that are enough to prevent disease in most vaccinated individuals. In countries where the use of vaccination is widespread, there has been a dramatic reduction in the number of people who become ill and die from formerly common and severe infections.3 For example, the whooping cough vaccine prevents disease in 85% of recipients, while the measles vaccine prevents disease in 95% of recipients from the first dose alone.48 The remaining proportion of individuals may not be fully protected and remain at least partially susceptible to infection. This may be due to genetic factors, or to the presence of other medical conditions that affect their ability to mount a protective immune response to the vaccine. These people are also protected when herd immunity has been achieved (see ‘The community at large benefits’ below).
Booster doses of some vaccines are required to maintain protection
Examples of vaccines that require booster doses include whooping cough, tetanus, and polio vaccines, as well as conjugate pneumococcal and meningococcal vaccines. In contrast, a single course of others, such as the hepatitis B vaccine, appears to be sufficient to provide lifelong protection.
After the first immunisation, a small and brief response occurs. When additional (booster) doses are given, memory lymphocytes created during the initial response are switched on to generate a much more rapid and longer lasting protective response. The graphic below shows the levels of antibodies from B cells after first and booster vaccinations.17
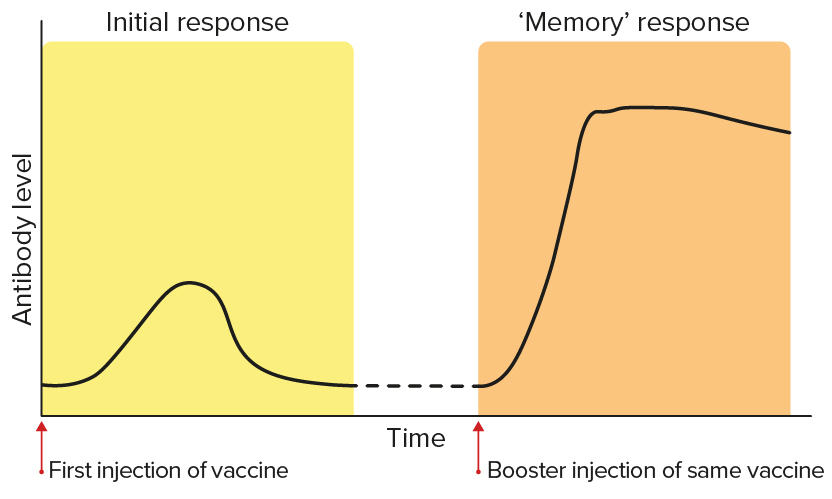
The levels of antibodies from B cells after first and booster vaccinations.
Vaccines can protect against long-term complications of infections
Encephalitis
Encephalitis is inflammation of the brain.
By preventing infection, vaccines can also prevent long-term complications associated with chronic infections, where the pathogen persists in the body after the initial infection has passed.
Certain viruses can cause dormant infections that persist in the body for years and cause long-term problems. Such persistent infections can eventually lead to chronic damage to infected organs (such as encephalitis induced by measles, or cirrhosis of the liver caused by hepatitis B or hepatitis C virus infection).49–51
Persistent viral infections can also lead to late complications such as cancer. For example, hepatitis B can lead to liver cancer and liver damage, and human papilloma virus can cause cancers including cervical and anal cancers.52–54 Vaccines are available to protect against these diseases.
Even if an individual has already been exposed to a disease, vaccines against that disease can still be beneficial in some cases, such as the chickenpox vaccine. This vaccine protects against the development of a long-term complication of the infection, shingles. Shingles is a debilitating condition characterised by the appearance of painful blisters on parts of the skin above nerves where the chickenpox virus has lain dormant since infection in childhood. Adults who had chickenpox in childhood can be given a high-dose chickenpox vaccine to boost immunity, resulting in a substantial reduction in their risk of developing shingles in the future. This is typically offered to elderly people, who are at higher risk of complications from shingles.55
The community at large benefits
Herd immunity occurs when a significant proportion of individuals within a population are protected against a disease through immunisation. This offers indirect protection for people who are still susceptible to the disease, by making it less likely that they will come into contact with someone who is carrying the pathogen.
Most vaccines not only benefit the individual who receives the vaccine but the entire population through a phenomenon called herd immunity.2
Herd immunity occurs when a significant proportion of individuals within a population are protected against a disease through immunisation. This situation offers indirect protection for people who are still susceptible to the disease, by making it less likely that they will come into contact with someone who is carrying the pathogen. Vaccination is the best way to create the right conditions for herd immunity to develop.
As well as protecting unvaccinated individuals, herd immunity benefits the small proportion of people who fail to respond adequately to vaccination or are unable to be vaccinated for medical reasons.56
In the case of a highly infectious disease such as measles, more than 95% of the population must be vaccinated to achieve sufficient herd immunity to prevent transmission if the disease recurs.
Vaccines across the life span

Newborn babies
The body’s immune system begins developing before birth. In the period during and soon after birth, when the functions of the immune system are still maturing, newborns are protected against many, but not all, serious infections by antibodies from their mother. This protection usually lasts for about four months.
Babies and children
As the early protection provided by the mother’s antibodies fades away, a child’s innate and adaptive immune systems start to mature and develop a memory of infections.57 They are more vulnerable to some infections at this age as their immune system is still maturing. However, their protection against pathogens grows throughout this time.
Children also have protection from early vaccinations. The current immunisation programs are designed to balance the ability of the child’s immune system to respond to the vaccine against the risk of the child getting the infection.
Adults
Adults require booster doses of some vaccines, such as tetanus, to maintain adequate levels of immunity throughout their life. Additional vaccines may be required in adulthood if the person plans on travelling overseas.
Not every person has a normally-functioning immune system, and some have primary or secondary immune deficiencies. A primary immune deficiency is one a person is born with, and a secondary immune deficiency is one that a person acquires from a disease or treatment, such as chemotherapy. Some people may be immunosuppressed from a disease or treatment, such as people receiving treatment for autoimmune diseases or cancer. These people may not be able to produce a strong immune response following vaccination and may rely on herd immunity to be protected from acquiring diseases.
Pregnancy
Maternal antibodies
Maternal antibodies are antibodies produced by a mother and transferred to the fetus.
Maternal antibodies cross the placenta into the baby’s circulation before birth and are also present in the mother’s breast milk. If the mother has been vaccinated or has recovered from infection during pregnancy, the amount of antibodies transmitted to the baby can be sufficient to ensure complete protection. On the other hand, if the mother’s infection (particularly with the pathogen that causes whooping cough) or immunisation occurred a long time ago, the antibody levels and protection may be lower.
Some vaccines, including inactivated vaccines, are considered safe to be given during pregnancy. The rates of side effects among pregnant women are similar to those in the general population. No link has been established between vaccination with inactivated vaccines in pregnancy and birth defects. The use of inactivated vaccines in pregnancy is particularly desirable for infections such as influenza and whooping cough that affect pregnant women or their babies more severely than the general population. This is because vaccination during pregnancy protects the mother against infection and provides protection to the baby as a result of the transfer of maternal antibodies.
Live attenuated vaccines, such as the MMR vaccine, are not recommended during pregnancy, as the live viruses could theoretically be transmitted from pregnant mothers to their baby. However, there is no evidence of increased birth defects in children whose mothers inadvertently received live attenuated vaccines while pregnant.
Older adults
Older adults, particularly people over 65, will experience a progressive decline in immunity as they get older. This means that they are more susceptible to infection and less responsive to vaccines.
Infection due to influenza, varicella-zoster viruses (causing chickenpox and shingles), SARS-CoV-2 (causing COVID-19) and Streptococcus pneumoniae can cause severe illness in older adults. Most of these diseases also lead to increased risk of death in older adults.
Special vaccines designed to work better with the immune system of older adults are made in two main ways:
- Developing new types of adjuvants to stimulate the immune response more effectively
- Increasing the amount of antigen included in the vaccine
Vaccines can control, eliminate and eradicate diseases
When a large proportion of a community is immunised, it can lead to a situation where there are very low disease levels in that population. This is referred to as control of the disease.
Even more effective and prolonged vaccination programs can result in interruption of transmission in the population for long enough to eliminate the disease. However, even when high levels of community coverage with a vaccine are achieved, infection may be reintroduced, such as by unvaccinated travellers. In Australia, isolated outbreaks of infectious diseases such as measles have been attributed to transmission from unvaccinated travellers.
Once a high degree of control is achieved worldwide, it is theoretically possible to eradicate an organism and the associated risk of infection, provided there is no other animal that can carry the infection and transmit it back to humans. This was achieved with smallpox in the 1970s. There is hope that such a goal may also be achievable for polio and measles, since (as with smallpox) humans are the only hosts. Of the three strains of polio, two strains have already been eradicated. Compared with 350,000 cases in 1988, only 33 polio cases were reported worldwide in 2018.
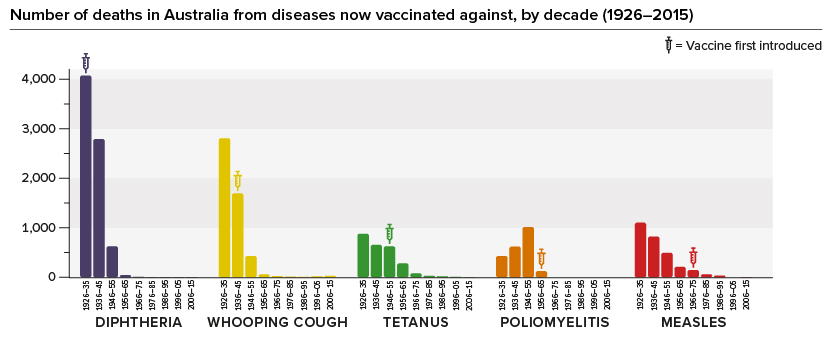
Vaccination brings economic benefits
In the first six years after the introduction of the polio vaccine worldwide:
- more than 150,000 cases of paralytic polio were prevented
- more than 12,500 deaths from polio were prevented
- more than US$30 billion per year (in 1999 dollars) was saved.58
The cost-effectiveness of community immunisation programs is measured by determining the benefits that result from preventing illness, disability and death, and comparing them with the costs of vaccine production and delivery to the population.
Better health, hygiene and vaccination reduces infections
Hygiene and healthcare improvements, such as widespread availability of antibiotics and better overall medical support systems, have reduced deaths from all diseases. However, vaccines have had an additional substantial impact: deaths in Australia from diphtheria, whooping cough, tetanus, polio and measles have all dramatically declined or disappeared since vaccines were introduced.
The introduction of the Haemophilus influenzae type b (Hib) vaccine in 2003 and the meningococcal type C vaccine in 2004 led to a very rapid and noticeable decline in the number of severe and sometimes fatal infections. This effect cannot be attributed to any change in living conditions or medical treatment.
Explore more
Watch the video
Read additional information
2. What is in a vaccine?

Ingredients
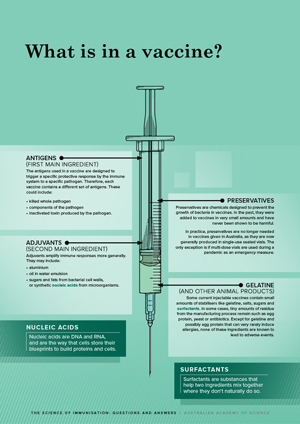
Antigens (first main ingredient)
The antigens used in a vaccine are designed to trigger a specific protective response by the immune system to a specific pathogen.17 Therefore, each vaccine contains a different set of antigens.28–30 These could include:
- killed whole pathogen
- components of the pathogen
- inactivated toxin produced by the pathogen.
Adjuvants (second main ingredient)
Adjuvants amplify immune responses more generally. They may include:
- aluminium
- oil in water emulsion
- sugars and fats from bacterial cell walls, or synthetic nucleic acids from microorganisms.
Preservatives
Nucleic acids
Nucleic acids are DNA and RNA, and are the way that cells store their blueprints to build proteins and cells.
Preservatives are chemicals designed to prevent the growth of bacteria in vaccines. In the past, they were added to vaccines in very small amounts and have never been shown to be harmful.2,31
In practice, preservatives are no longer needed in vaccines given in Australia, as they are now generally produced in single-use sealed vials. The only exception is if multi-dose vials are used during a pandemic as an emergency measure.
Surfactants
Surfactants are substances that help two ingredients mix together where they don’t naturally do so.
Gelatine (and other animal products)
Some current injectable vaccines contain small amounts of stabilisers like gelatine, salts, sugars and surfactants.32,33 In some cases, tiny amounts of residue from the manufacturing process remain such as egg protein, yeast or antibiotics. Except for gelatine and possibly egg protein that can very rarely induce allergies, none of these ingredients are known to lead to adverse events.
Several different types of antigen are used in vaccines
Some vaccines contain the killed whole microorganism that the vaccine is designed to protect against. The virus or bacterium is grown in a laboratory and killed by heat or chemicals so that it is no longer infectious.34 The injectable polio vaccine and inactivated hepatitis A vaccine are examples of this type.
Other vaccines contain only components of the pathogen as their antigens. These components can be prepared by purifying them from the whole bacterium or virus, or by genetically engineering them.35–37 Engineered vaccines include the human papillomavirus vaccine, which protects against cervical cancer, and the hepatitis B virus vaccine.
In some vaccines, components of the pathogen are linked with proteins to create an antigen that can generate a stronger response—this allows even 6-week-old babies to make significant amounts of antibodies, which they otherwise could not do until they are older.38 These vaccines are called conjugate vaccines and include those against Haemophilus influenzae type b (Hib) infection, meningococcal and pneumococcal disease.
Another group of vaccines is based on the toxin produced by the pathogen that causes the disease symptoms. The toxin is chemically treated to make it harmless. The antibodies produced against it can still neutralise the toxin and prevent disease symptoms from developing. Examples of this type include tetanus and diphtheria vaccines.
Some vaccines contain live organisms
Attenuation
The attenuation process permanently alters the pathogen so that it is still able to reproduce and stimulate an immune response, but does not cause disease.
Some vaccines contain an infectious microorganism; these are called live vaccines. The microorganism may be derived from the pathogen that the vaccine aims to protect against. This is usually achieved by growing the pathogen in the laboratory under conditions designed to weaken or attenuate it.39 Examples include the injectable MMR vaccine and the chickenpox vaccine.
Alternatively, a live vaccine may consist of a naturally occurring organism closely related to the pathogen that does not cause disease in healthy humans with intact immune systems. An example is the BCG vaccine against tuberculosis and leprosy.
Vaccines containing live pathogens are not recommended for people whose immune systems are impaired due to the use of immunosuppressive drugs, serious illness or abnormalities of the immune system, because of the risk of causing disease. Similarly, live vaccines are not recommended during pregnancy as a precautionary measure, in case the pathogens they contain cross the placenta to the unborn baby. This is because a baby’s immune system is not completely developed until after birth. Vaccines without live microorganisms (inactivated vaccines), in contrast, have not been shown to be harmful in pregnancy.
Adjuvants amplify the immune system’s response
Adjuvants are not needed in vaccines that use live organisms, as these naturally produce inflammation and amplify protective immunity.
Adjuvants are substances that promote a more vigorous immune response to vaccine antigens. They can also help target the body’s response. In doing so, they may cause mild local reactions (soreness, redness and swelling) at the injection site. These reactions are expected and part of the innate immune response.
Most inactivated vaccines use adjuvants to make the body’s defences think a significant infection is present. They stimulate stronger, longer-lasting immune responses to the vaccine antigens, leading to better protection against subsequent infection. Adjuvants are not needed in vaccines that use live organisms, as these naturally produce inflammation and amplify protective immunity.
Aluminium salt (known as alum) has been frequently used in human vaccines as an adjuvant and has a track record of safety dating back to the 1950s.40 Some newer vaccines incorporate more active adjuvants, derived from naturally-occurring oil in water emulsions (mixtures), fats or sugars from bacterial cell walls, or synthetic nucleic acids mimicking bacterial DNA. These can produce more vigorous and better-targeted immune responses against the infectious agent.41
Vaccine quality is carefully monitored
In addition to adjuvants and antigens, vaccines can contain tiny quantities of materials from the manufacturing process. These can include trace amounts of detergents, nutrients from the laboratory cultures, chemicals used to kill the pathogens, stabilisers like gelatine, or small amounts of DNA and parts of dead organisms.
Vaccine developers are required by regulatory authorities to test for the presence of these extra materials during the manufacturing process to ensure they do not exceed levels known to be safe.
Occasionally, individuals can be allergic to a vaccine ingredient, although such reactions are rare. Reviews of vaccine monitoring data have shown that less than 1 out of 100,000 people might experience a severe allergic reaction.42,43 Vaccines do not cause allergic diseases.
Vaccines contain trace amounts of DNA
The presence of DNA or RNA (genetic instructions) in a vaccine does not lead to changes to the DNA or RNA of the person who receives it.
Because most vaccine antigens are prepared from whole organisms, a vaccine may contain some of that organism’s genetic material in the form of DNA, or a similar type of molecule known as RNA. The amount of genetic material in a vaccine is minuscule, much less than the amount we eat in our food each day.44 Vaccines based on living pathogens contain that organism’s genetic information because it is necessary for the vaccine to work. However, the DNA (or RNA) in the pathogen does not remain for long or lead to long-term detrimental effects in the vaccinated person.45 The presence of DNA or RNA in a vaccine does not lead to changes in the vaccinated person’s DNA or RNA.
Some vaccines must be given with caution to people with a history of allergies to eggs or red meat
Some vaccines, such as influenza or MMR vaccines, contain antigens from viruses grown in eggs or on chick cells and may contain some egg proteins. However, newer MMR vaccines contain so little egg protein that it is now considered safe to give them even to someone who is already known to be very sensitive to egg protein.46 The seasonal influenza vaccines in current use contain minimal amounts of egg protein and can be used in most egg‑sensitive people.47
The viruses in two other less-frequently-used vaccines (for Q fever and yellow fever) are also grown in eggs, and specialist advice should be sought if either of these vaccines are needed for a person with severe egg allergy.
Small amounts of gelatine of animal origin are used to stabilise some vaccines like Zostavax and MMR. Allergic reactions to gelatine have been reported, but are rare and usually mild in recipients of these two vaccines. However, in people with mammalian meat allergy, the chance of serious anaphylactic reactions is increased. Specialist advice should be sought in such cases before administering a vaccine like Zostavax, the chickenpox and shingles vaccine.
Fetal tissue cells may be used during vaccine development
Fetus
Fetus is a term used to describe a baby before they are born.
Certain viruses grown for use in vaccines require the use of ‘cell lines’. These cell lines were derived over 40 years ago from human fetal tissue. Vaccines manufactured using these cell lines include those for rubella, chickenpox, shingles and hepatitis A. Copies of these cells are still used because they are excellent viral factories and are free of contaminants, which can be a problem when viruses are grown in cells that are not from humans. Once the cell has been used to grow the virus, the virus is taken out and purified—the human cells are not included in the vaccine. It is estimated that the vaccines made using cells from these original fetal tissue samples have prevented nearly 11 million deaths and prevented at least 4 billion cases of disease.
Vaccines are mostly injected into muscle or fat
Vaccines are commonly given by injection into a muscle—this is called intra-muscular injection. Some vaccines can be injected into the layer of fat just under the skin—this is called subcutaneous injection. Some vaccines can be given orally (through the mouth) instead of as an injection, for example the oral polio vaccine.
Vaccination sets off a chain of events resulting in movement of immune system cells to the area where the vaccine was administered, then through to the lymph nodes and spleen, eventually leading to the development of immunity throughout the body. Vaccines are not delivered directly into the bloodstream. One common side effect of intramuscular injection is some short-term discomfort at the injection site. Severe side effects, however, are very rare.

Vaccination sets off a chain of events resulting in movement of immune system cells to the area where the vaccine was administered, then through to the lymph nodes and spleen, eventually leading to the development of immunity throughout the body. Image: Self Magazine CC-BY-2.0
Explore more
Watch the video
Read additional information
1. What is immunisation?

Immunisation protects against infectious disease
The purpose of immunisation is to prevent people from getting sick. It helps to protect people against the complications of becoming ill, including developing chronic diseases, cancer, and death.6–8
Vaccines work by stimulating the body’s defence mechanisms to provide protection against infection.
Vaccines can sometimes produce a stronger, longer-lasting protective response compared to immunity from a natural infection.
Vaccines create immunity without causing disease. Disease can lead to serious complications, which is why vaccination is a safer way to develop immunity.
Vaccines work by stimulating the body’s defence mechanisms to provide protection against infection and illness. These defence mechanisms are collectively referred to as the immune system. Vaccines mimic and sometimes improve the protective response normally mounted by the immune system after infection. The great advantage of immunisation over natural infections is that immunisation has a much lower risk of harmful outcomes.2–4,9–15
Immunisation harnesses the body’s own defence mechanisms
To understand how immunisation protects against the diseases produced by pathogens (such as viruses and bacteria), we first need to understand how the immune system works.16,17

The immune system
The immune system is the body’s defence mechanism, protecting against invaders like bacteria and viruses to keep us healthy.
Cells are the main building blocks of our body. Our immune system relies on many different types of cells, each playing an important role. Many of these can be found in our bloodstream, especially white blood cells, which are the main component of the human immune system.
White blood cells are strategically located throughout the body, not only in the bloodstream but in the lymph nodes, spleen, lungs, intestines and skin. This allows them to deal with pathogens wherever they enter the body.
There are two main types of white blood cells:
- guardian cells responsible for innate immunity (your body’s first defence against pathogens)
- lymphocytes responsible for specific or ‘adaptive’ immunity (your body's ability to remember pathogens and react quickly if re-infected).
Other blood cell types include red blood cells, which carry oxygen to our tissues, and platelets, which help our blood to clot.
Innate immunity
The skin and the lining of the lungs and intestine are the first line of defence against infection, forming a physical barrier for protection. These tissues and the sentinal cells that live there form the innate immune system.17,18
Some of these cells ingest pathogens or vaccine particles and use these to activate lymphocytes (part of specific immunity).
Some innate immunity cells produce chemicals capable of causing inflammation and amplifying the response of specific immunity.
The innate immune system gives a generalised response towards anything it identifies as ‘foreign’. By itself, that response might not be strong enough to protect against an infection.
Specific immunity
After a person has an infection or is vaccinated, specific lymphocytes learn to recognise their target antigens and multiply. Some then become effector cells that can eliminate or prevent infection, while others turn into long-lived memory cells that are ready to respond more rapidly and effectively if the infection returns. They are ‘specific’ because they are created to target and respond only to that antigen.
There are two types of lymphocytes: T cells and B cells.19–21
- T cells respond to infections by releasing chemicals called cytokines, which trigger protective inflammation. T cells can also kill cells that have a pathogen, such as a virus, hidden inside them.22
- B cells, often with help from T cells, are involved in making antibodies. Antibodies are complex proteins that attach in a ‘lock-and-key’ fashion either to pathogens or to the toxins released by them.
When antibodies attach to a pathogen they flag it for destruction, and when they attach to a toxin they neutralise its ability to cause damage. 16,17
The immune system’s responses to pathogens stops the infection in most cases, followed by repair of any damage to the body. However, serious infections can overwhelm the immune system’s capacity to respond and can lead to severe disease or death. Giving a vaccine before exposure to the infection generates protective immunity in advance and avoids the serious outcomes of the disease.
Vaccination is disease-specific
Immune responses are very specific, and that is the reason we need to have a specific vaccine for each disease.
The immune system responds separately to each pathogen it encounters. It cannot be ‘overloaded’ by giving the full range of currently available vaccines or by having multiple antigens in one vaccine.
A healthy immune system can generate hundreds of millions of T and B cells, each of which targets one particular antigen.23
However, pathogens can sometimes overwhelm the immune response. Vaccines give the immune system a head start by allowing it to learn and remember what a pathogen looks like, providing valuable protection against aggressive pathogens.
These immune responses are very specific, so we need to have a separate vaccine for each disease. The immune system can respond independently to each pathogen it encounters. This is why the system cannot be ‘overloaded’ or damaged by giving the full range of currently available vaccines or by having multiple antigens in one vaccine.
Vaccines work with the immune system’s ability to remember
When the immune system recognises a pathogen, individual lymphocytes make antibodies and cytokines against the infection and multiply quickly. As a result, the number of lymphocytes (T and B cells) specific for that infection increases, enabling the body to fight the infection more efficiently.
Most of the cells involved in immune responses live for only a few days, but a small number of lymphocytes survive for months or years after the infection has been cleared away. These lymphocytes either continue to produce antibodies or retain a ‘memory’ of the invading pathogen.17,24,25 In the case of measles, for example, that memory has been shown to last for more than 60 years.26
The way the immune system remembers infections is one of its most valuable assets. This memory means the immune system can mount a much faster, larger and more sustained response if it encounters the same pathogen again.17,24,25 That response can control subsequent infection more efficiently, without leading to the unwanted and serious complications associated with the infection itself.
Newborn vaccines work with the newborn immune system
The body’s immune system begins developing before birth.27 A mother’s antibodies protect newborns against many (but not all) serious infections during and soon after birth, while the baby’s immune system function is still maturing. This protection usually lasts for about four months.
The current immunisation programs for infants are designed to balance waiting for the baby’s immune system to have the capacity to respond to the vaccine, against the risk of the baby getting an infection.
Chronic carrier
Chronic carriers are people who remain hosts of pathogens for months or years after they were first infected.
In the case of hepatitis B, for example, the risk to the baby is high, and exposure to the virus at birth or in the first few months of life can result in the infant becoming a chronic carrier for life. That is why vaccination for hepatitis B starts within one week of birth.
The situation is different for other pathogens, either because there is a lower risk of infection in the first few months of life or because they would not produce a protective immune response to them at that age. For example, administering the vaccines against Haemophilus influenzae type b (Hib) and Streptococcus pneumoniae is delayed until 6–8 weeks of age when the infant’s immune system can respond more effectively. The measles-containing MMR (measles, mumps and rubella) vaccine is not given until 12 months of age, when maternal antibodies against measles, which can interfere with vaccine responses, have essentially disappeared.
Pre-formed antibodies provide immediate protection
It takes around 7–21 days after being vaccinated to generate an effective immune response in healthy individuals.
Most vaccines work by switching on a person’s immune system to make the antibodies, cytokines and memory cells needed to protect against infection. However, this kind of active immune response takes 7–21 days to fully develop.
Sometimes, in the case of overwhelming and dangerous infections, an unwell person may receive pre-formed antibodies as part of their medical treatment to prevent or combat the infection. These either come from healthy blood donors or are produced in a laboratory, as they can act much more quickly to help the person fight off the infection.17 This is known as ‘passive immunisation’. However, these antibodies don't stay in the body for very long—it is better to make antibodies through being vaccinated wherever possible.
It takes around 7–21 days after being vaccinated to generate an effective immune response in healthy individuals. Image: National Centre for Immunisation Research and Surveillance






